 |
 |
| Tuberc Respir Dis > Volume 78(2); 2015 > Article |
|
Abstract
We report a case of agranulocytosis caused by ethambutol in a 79-year-old man with pulmonary tuberculosis. He was referred for fever and skin rash developed on 21th day after antituberculosis drugs (isoniazid, rifampicin, ethambutol, and pyrazinamide) intake. Complete blood count at the time of diagnosis of pulmonary tuberculosis was normal. On the seventh admission day, agranulocytosis was developed with absolute neutrophil count of 70/┬ĄL. We discontinued all antituberculosis drugs, and then treated with granulocyte colony-stimulating factor. Three days later, the number of white blood cell returned to normal. We administered isoniazid, pyrazinamide, and ethambutol in order with an interval. However, fever and skin rash developed again when adding ethambutol, so we discontinued ethambutol. After these symptoms disappeared, we added rifampicin and ethambutol in order with an interval. However after administering ethambutol, neutropenia developed, so we discontinued ethambutol again. He was cured with isoniazid, rifampicin, and pyrazinamide for 9 months.
Antituberculosis drugs may induce hematologic side effects such as thrombocytopenia and neutropenia, but agranulocytosis has rarely been reported. We describe a patient who developed agranulocytosis induced by ethambutol.
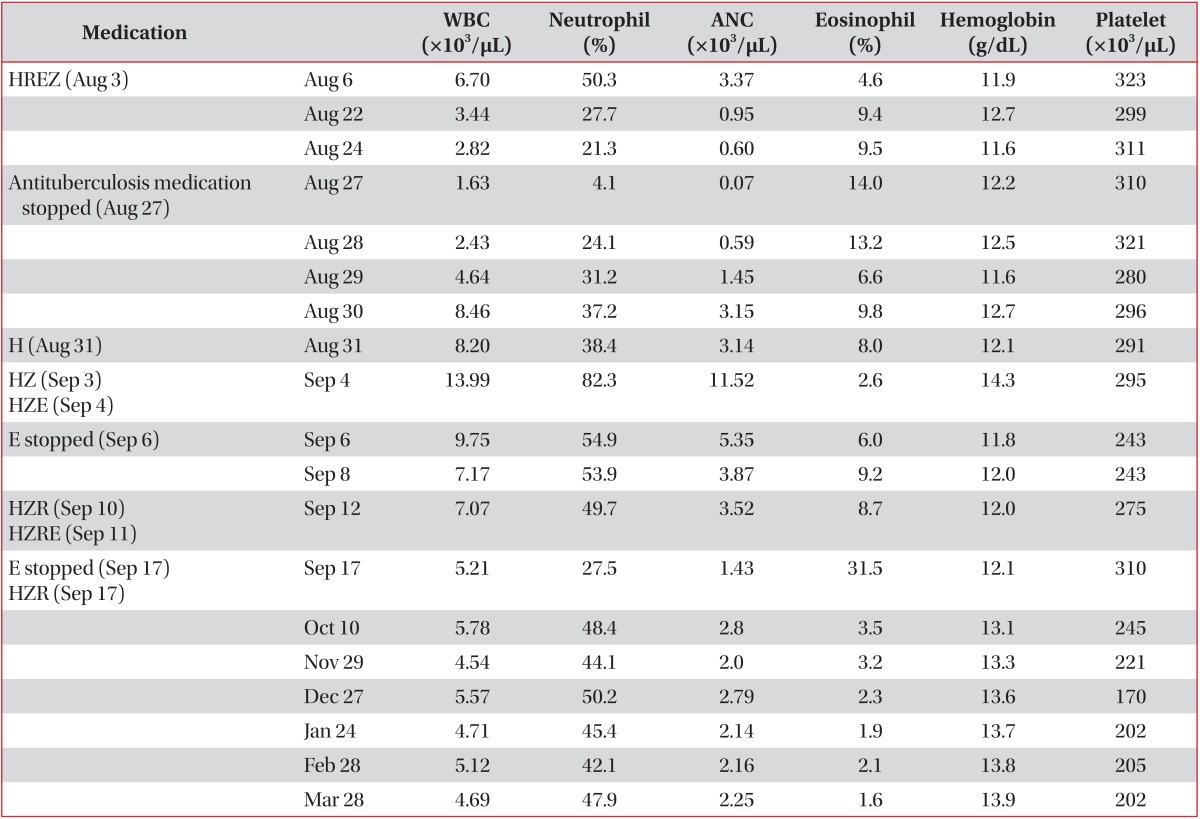
A 79-year-old man was referred to our hospital for fever and skin rash showed no further improvement with antipyretic drug and fluid therapy on 3 weeks after taking antituberculosis drugs (isoniazid, rifampicin, ethambutol, and pyrazinamide) at other hospital. Three weeks ago, he was diagnosed with pulmonary tuberculosis by positive acid-fast bacilli. Complete blood count (CBC) at the time of diagnosis of pulmonary tuberculosis was white blood cell (WBC) of 6.70├Ś103/┬ĄL, absolute neutrophil count (ANC) of 3.37├Ś103/┬ĄL, hemoglobin of 11.9 g/dL, and platelet of 323├Ś103/┬ĄL. He denied other medical disease except the known pulmonary tuberculosis.
On the seventh admission day, agranulocytosis was developed with ANC of 70/┬ĄL. The patient had WBC of 1.63├Ś103/┬ĄL, hemoglobin of 12.2 g/dL, and platelet of 310├Ś103/┬ĄL. We stopped all antituberculosis drugs, and then treated with granulocyte colony-stimulating factor, subcutaneous dose of 300 ┬Ąg of biosimilar filgrastim. Three days later, the number of WBC was 8.46├Ś103/┬ĄL and the number of ANC was 3.15├Ś103/┬ĄL (Figure 1). And then he was administered only isoniazid again for 3 days. Since there was no adverse effects, we added pyrazinamide and ethambutol, respectively with an interval. However, we stopped ethambutol due to fever and skin rash arised after adding ethambutol. There were no adverse effects of isoniazid and pyrazinamide for several days, so we added rifampicin and added ethambutol again with an interval. However, due to relapse of neutropenia (ANC of 1.43├Ś103/┬ĄL) in the sixth day after administrating ethambutol, we stopped ethambutol. And we continued isoniazid, rifampicin, and pyrazinamide for 9 months. He was cured without any other hematologic disorders.
Judging from the clinical course, we could conclude that ethambutol had caused agranulocytosis (Table 1).
Antituberculosis drugs have been shown to produce adverse effects from insignificance to life-threatening. In an immunocompetent patient, neutropenia is rare adverse effects of antituberculosis therapy. However, neutropenia combined with sepsis could lead to life-threatening. Umeki1 reported that the incidence rate of neutropenia and agranulocytosis due to antituberculosis drugs were observed in 10.9% and 3.96%, respectively. Shishido et al.2 reported that the incidence rate of agranulocytosis due to antituberculosis drugs was estimated at 0.06%. Therefore, it is important to detect neutropenia earlier and treat it in a patient taking antituberculosis drugs.
The causes of agranulocytosis mentioned above1,2 were not ethambutol, but rifampicin and isoniazid. The important adverse effects of ethambutol are visual disturbance due to retrobulbar neuritis and toxicities to peripheral nerves3,4. There is no well-documented report of agranulocytosis induced by ethambutol. Though Wong and Yew5 suggested an association of neutropenia (ANC of 1.4├Ś103/┬ĄL) with ethambutol, but it was not agranulocytosis, but mild neutropenia.
Our case is the first report of agranulocytosis induced by ethambutol. According to the previous reports1,2,6,7, neutropenia can be developed in a patient who is treated with antituberculosis drugs during a few months after starting administration. In administrating antituberculosis drugs including ethambutol, as in our case, we should regularly make a close monitoring of the CBC to avoid serious events.
References
1. Umeki S. Adverse effects of antitubercular drugs and significance of measurement of the drug-stimulating lymphocyte transformation rate. Jpn J Med 1989;28:335-340. PMID: 2739142.


2. Shishido Y, Nagayama N, Masuda K, Baba M, Tamura A, Nagai H, et al. Agranulocytosis due to anti-tuberculosis drugs including isoniazid (INH) and rifampicin (RFP): a report of four cases and review of the literature. Kekkaku 2003;78:683-689. PMID: 14672045.

3. Citron KM. Ethambutol: a review with special reference to ocular toxicity. Tubercle 1969;50(Suppl):32-36. PMID: 4918612.
5. Wong CF, Yew WW. Ethambutol-induced neutropenia and eosinophilia. Chest 1994;106:1638-1639. PMID: 7956448.


6. Tattersall PE. Agranulocytosis and anaemia associated with disseminated tuberculosis. Br Med J 1957;2:1351-1352. PMID: 13479719.



7. Jenkins PF, Williams TD, Campbell IA. Neutropenia with each standard antituberculosis drug in the same patient. Br Med J 1980;280:1069-1070. PMID: 7388400.



- TOOLS
-
METRICS

- Related articles
-
Prednisolone in The Treatment of Pulmonary Tuberculosis
Cost as a Factor in the Selection of Patients for Operation in Pulmonary Tuberculosis
Surgical Treatment in Pulmonary Tuberculosis
Study on the Candida in Patients with Pulmonary Tuberculosis
A Study on Hospitalized Patients with Pulmonary Tuberculosis1990 March;37(1)



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation



