 |
 |
| Tuberc Respir Dis > Volume 84(2); 2021 > Article |
|
Abstract
Background
Methods
Results
Notes
AuthorsŌĆÖ Contributions
Conceptualization: Kim EJ. Methodology: Kim EJ, Hong HL. Formal analysis: Kim EJ. Data curation: Kim EJ, Choi KJ. Software: Kim EJ. Validation: Hong HL. Investigation: Choi KJ, Kim EJ. Visualization: Kim EJ, Choi KJ. Writing - original draft preparation: Kim EJ. Writing - review and editing: Choi KJ, Hong HL, Kim EJ. Approval of final manuscript: all authors.
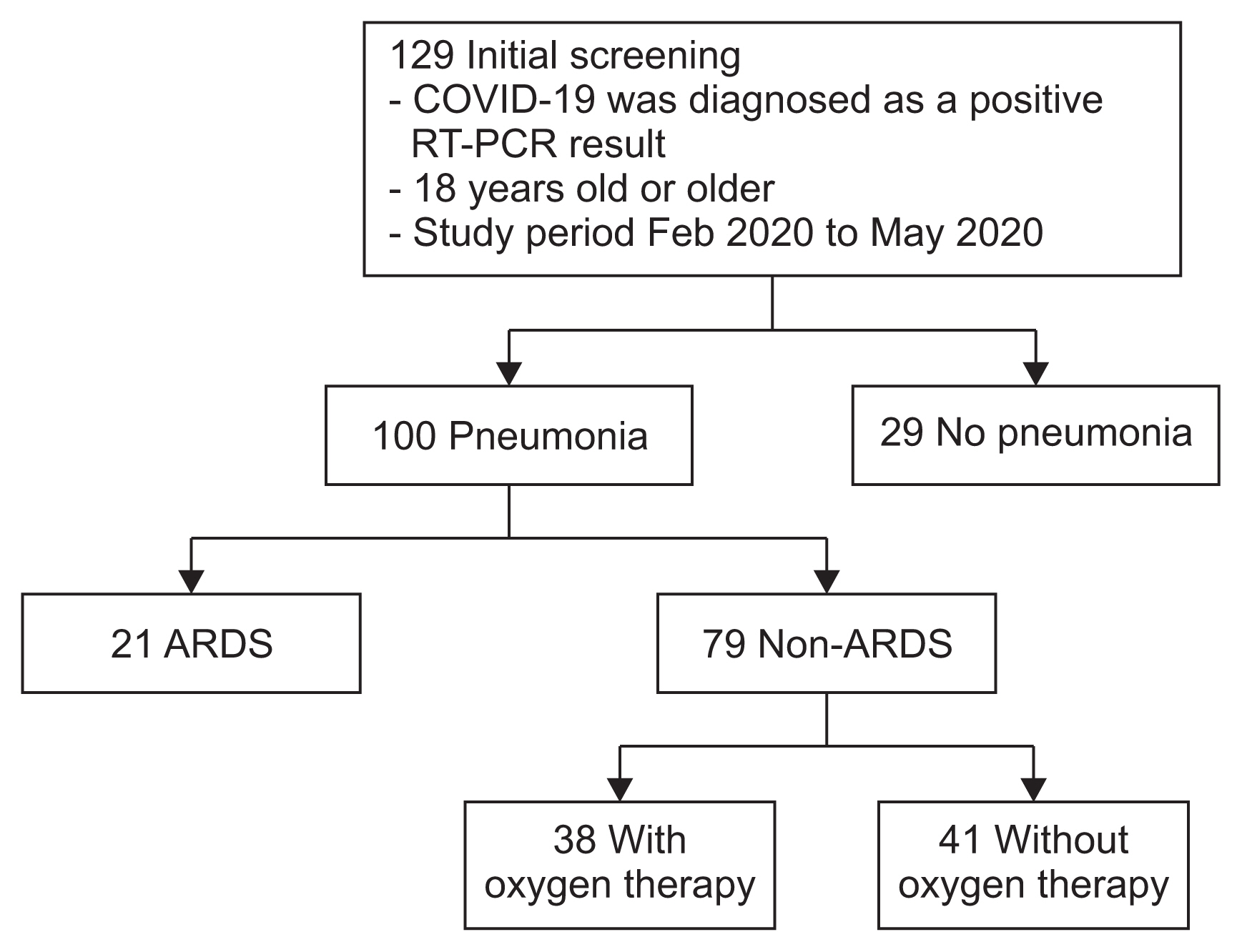
Figure┬Ā1
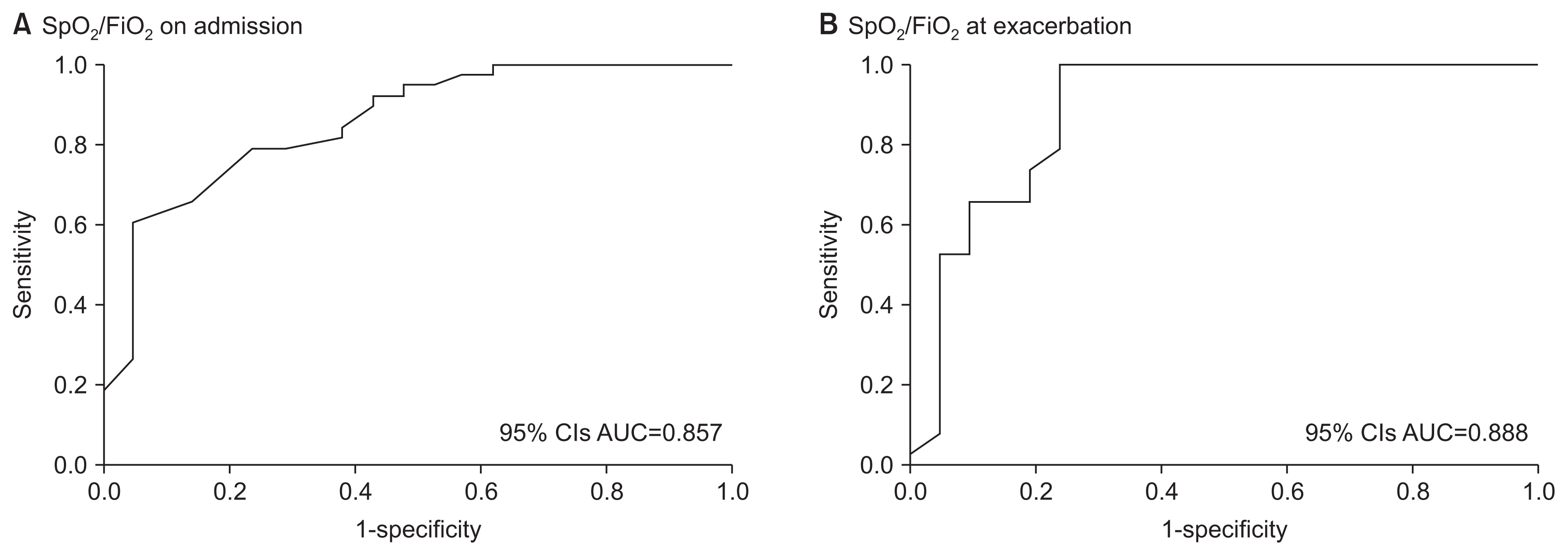
Figure┬Ā2
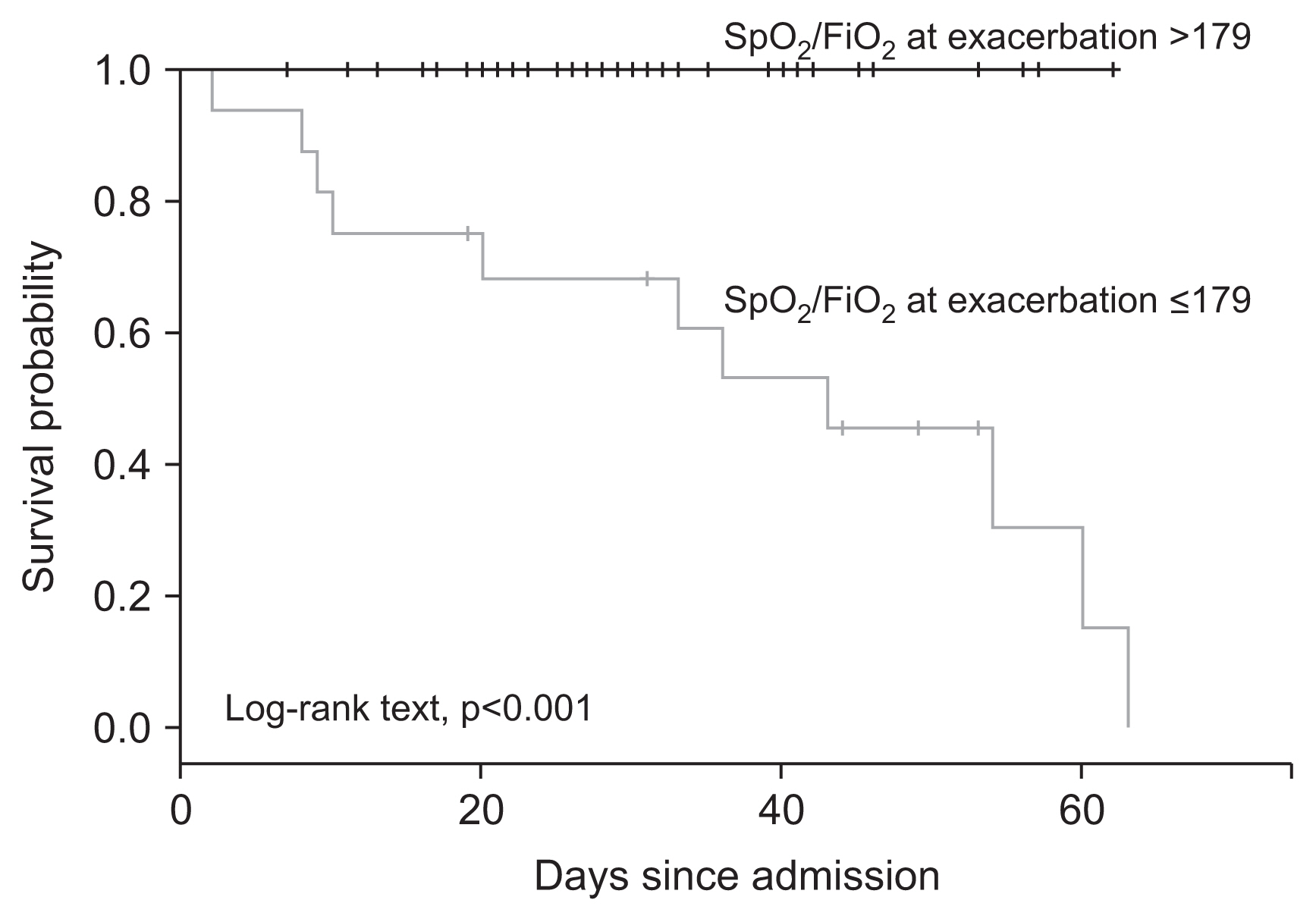
Figure┬Ā3
Table┬Ā1
| Characteristic | ARDS (n=21) | Non-ARDS pneumonia requiring oxygen (n=38) | p-value |
|---|---|---|---|
| Age, yr | 75.0 (63.0-81.0) | 70.0 (58.0-79.0) | 0.254 |
| Sex (male:female) | 13:8 | 8:30 | 0.002* |
| Comorbidity | 18 (85.7) | 28 (73.7) | 0.286 |
| ŌĆāDiabetes mellitus | 6 (28.6) | 10 (26.3) | 0.852 |
| ŌĆāHypertension | 9 (42.9) | 15 (39.5) | 0.800 |
| ŌĆāPulmonary disease | 6 (28.6) | 7 (18.4) | 0.368 |
| ŌĆāLiver disease | 3 (14.3) | 2 (5.3) | 0.233 |
| ŌĆāHeart disease | 4 (19.0) | 6 (15.8) | 0.749 |
| ŌĆāKidney disease | 1 (4.8) | 4 (10.5) | 0.447 |
| ŌĆāMalignancy | 1 (4.8) | 1 (2.6) | 0.665 |
| ŌĆāPsychologic disease | 2 (9.5) | 3 (7.9) | 0.830 |
| ŌĆāCerebrovascular disease | 6 (28.6) | 9 (23.7) | 0.680 |
| ŌĆāImmuno-compromised condition | 1 (4.8) | 4 (10.5) | 0.447 |
| Smoking | 0.233 | ||
| ŌĆāNon-smoker | 18 (85.7) | 36 (94.7) | |
| ŌĆāCurrent smoker | 3 (14.3) | 2 (5.3) | |
| Laboratory tests | |||
| ŌĆāLeukocyte, ├Ś109/L | 6.1 (5.1-7.7) | 5.5 (3.8-7.1) | 0.217 |
| ŌĆāHematocrit, % | 37.8 (32.8-40.6) | 37.3 (34.5-39.7) | 0.740 |
| ŌĆāPlatelet, ├Ś103/mL | 177.0 (146.0-253.0) | 168.5 (141.0-267.0) | 0.722 |
| ŌĆāSegmented neutrophil, % | 80.5 (77.9-86.7) | 68.5 (60.6-79.4) | 0.002* |
| ŌĆāLymphocyte, % | 11.0 (7.5-14.4) | 20.0 (13.3-27.0) | 0.005* |
| ŌĆāEosinophil, % | 0.1 (0-0.2) | 0.5 (0.1-1.2) | 0.001* |
| ŌĆāCRP, mg/L | 83.0 (57.0-133.8) | 42.7 (23.3-78.2) | 0.002* |
| ŌĆāLDH, U/L | 415.0 (383.0-500.0) | 293.0 (229.5-339.5) | <0.001* |
| ŌĆāTotal bilirubin, mg/dL | 0.6 (0.4-0.7) | 0.5 (0.4-0.8) | 0.960 |
| ŌĆāTotal protein, g/dL | 6.6 (6.1-6.8) | 6.4 (6.1-6.8) | 0.787 |
| ŌĆāAlbumin, g/dL | 3.5 (3.3-3.8) | 3.7 (3.4-4.0) | 0.111 |
| ŌĆāBUN, mg/dL | 18.7 (13.9-24.2) | 15.2 (10.7-20.2) | 0.083 |
| ŌĆāCreatinine, mg/dL | 0.9 (0.7-1.2) | 0.8 (0.7-0.9) | 0.169 |
| ŌĆāSodium, mmol/L | 133.0 (130.0-137.0) | 138.0 (134.0-141.0) | 0.001* |
| ŌĆāProcalcitonin, ng/mL | 0.18 (0.10-0.30) | 0.09 (0.06-0.21) | 0.019* |
| ŌĆāTroponin-T, ng/mL | 0.01 (0.01-0.10) | 0.01 (0.01-0.01) | 0.136 |
| NEWS on admission | 7.0 (5.0-9.0) | 4.0 (3.0-6.0) | <0.001* |
| Time from symptom onset to 1st viral conversion (negative in RT-PCR), day | 25.0 (22.0-34.0) | 27.0 (22.0-34.0) | 0.692 |
| Time from symptom onset to second serial viral conversion (negative in RT-PCR), day | 32.0 (26.5-36.5) | 30.5 (26.0-39.0) | 0.886 |
| Hospital stay, day | 31.0 (20.0-44.0) | 28.5 (20.0-40.0) | 0.757 |
| No. of dead | 11 (52.4) | 0 | <0.001* |
| Treatment | |||
| ŌĆāInvasive mechanical ventilator | 10 (47.6) | 0 | <0.001* |
| ŌĆāHFNC | 15 (71.4) | 0 | <0.001* |
| ŌĆāECMO | 2 (9.5) | 0 | 0.053 |
| ŌĆāLopinavir/ritonavir | 18 (85.7) | 27 (71.1) | 0.205 |
| ŌĆāHydroxychloroquine | 15 (71.4) | 13 (34.2) | 0.006* |
| ŌĆāAntibiotics | 21 (100) | 36 (94.7) | 0.285 |
| ŌĆāSteroid | 18 (85.7) | 3 (7.9) | <0.001* |
Table┬Ā2
| ARDS (n=21) | Non-ARDS pneumonia requiring oxygen (n=38) | p-value | |||
|---|---|---|---|---|---|
|
|
|
||||
| No. | Median (IQR) | No. | Median (IQR) | ||
| SpO2 on admission, % | - | 91.0 (88.0-92.0) | - | 95.0 (92.0-96.0) | <0.001* |
|
|
|||||
| FiO2 on admission | - | 0.32 (0.21-0.60) | - | 0.21 (0.21-0.21) | <0.001* |
|
|
|||||
| PaO2 on admission, mm Hg | 11 | 78.7 (54.5-85.7) | 5 | 83.6 (66.1-110.0) | 0.583 |
|
|
|||||
| PF ratio on admission | 11 | 135.8 (111.3-262.2) | 5 | 398.1 (314.8-519.4) | 0.013* |
|
|
|||||
| SF ratio on admission | - | 287.5 (135.0-433.3) | - | 452.4 (438.1-457.1) | <0.001* |
|
|
|||||
| Time from start oxygen therapy to maintain to the highest FiO2, hr | 21 | 16.0 (9.5-46.0) | 20 | 27.0 (7.0-92.0) | 0.569 |
|
|
|||||
| SpO2 at exacerbation, % | 21 | 94.0 (90.0-98.0) | 20 | 96.5 (93.0-98.0) | 0.305 |
|
|
|||||
| FiO2 at exacerbation | 21 | 0.80 (0.70-0.90) | 20 | 0.30 (0.28-0.40) | <0.001* |
|
|
|||||
| PaO2 at exacerbation, mm Hg | 19 | 66.8 (55.1-84.0) | 2 | 56.1 (52.6-59.6) | 0.343 |
|
|
|||||
| PF ratio at exacerbation | 19 | 118.3 (78.5-153.0) | 2 | 179.5 (146.1-212.9) | 0.238 |
|
|
|||||
| SF ratio at exacerbation | 21 | 111.1 (102.2-139.0) | 20 | 319.0 (247.5-346.4) | <0.001* |
|
|
|||||
| Duration of oxygen therapy, day | - | 24.0 (13.0-40.0) | - | 19.0 (12.5-23.5) | 0.181 |
Table┬Ā3
| Univariate, unadjusted | Multivariate* | |||
|---|---|---|---|---|
|
|
|
|||
| HR (95% CI) | p-value | HR (95% CI) | p-value | |
| Sex | 2.222 (0.615-8.026) | 0.223 | - | - |
|
|
||||
| NEWS | 1.388 (1.145-1.684) | 0.001 | 1.277 (1.010-1.615) | 0.041 |
|
|
||||
| SF ratio on admission | 0.992 (0.987-0.997) | 0.001 | - | - |
|
|
||||
| SF ratio at exacerbation | 0.962 (0.923-1.002) | 0.065 | 0.916 (0.846-0.991) | 0.029 |
|
|
||||
| Neutrophil count | 1.073 (1.001-1.150) | 0.046 | 1.082 (0.995-1.177) | 0.066 |
|
|
||||
| Monocyte count | 0.763 (0.609-0.957) | 0.019 | - | - |
|
|
||||
| LDH | 1.002 (0.999-1.006) | 0.140 | 0.994 (0.998-1.000) | 0.050 |
|
|
||||
| Sodium | 0.813 (0.728-0.909) | <0.001 | - | - |
References
-
METRICS

- ORCID iDs
-
Eun Jin Kim

https://orcid.org/0000-0001-9791-8077Hyo-lim Hong

https://orcid.org/0000-0003-2147-1381Keum-Ju Choi

https://orcid.org/0000-0003-2282-9605 - Related articles


 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation



