Role of Single Port Rigid Thoracoscopy in Undiagnosed Pleural Effusion
Article information
Abstract
Background
In recent years, medical thoracoscopy has been well established to play an important role in undiagnosed pleural effusion; however, this procedure is underutilized due to limited availability of the instruments it requires. This study analysed the outcome of single port rigid thoracoscopy in patients with undiagnosed pleural effusions.
Methods
This study retrospectively analysed the outcomes of all patients with undiagnosed pleural effusion presenting to our centre between 2016 to 2020 who underwent single port rigid medical thoracoscopy as a diagnostic procedure.
Results
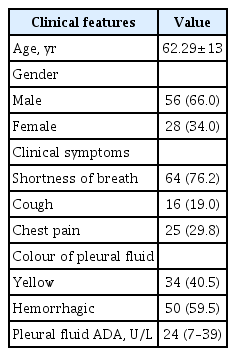
In total, 92 patients underwent single port rigid medical thoracoscopy. The most common presenting symptom was shortness of breath. A majority of the patients had lymphocytic exudative pleural effusion. The average biopsy sample size was 18 mm, and no major complication was reported in any of the patients.
Conclusion
Single port rigid thoracoscopy is a safe and well-tolerated procedure that yields a biopsy of a larger size with high diagnostic yield. Moreover, the low cost of the instruments required by this procedure makes it particularly suited for use in developing countries.
Introduction
Medical thoracoscopy, as an aspect of interventional pulmonology, is a minimal invasive procedure that provides highly sensitive and accurate results in undiagnosed pleural effusion. In clinical practice, pleural effusions are commonly seen with varying underlying aetiologies. However, the accurate diagnosis of such effusions depends on the population, geographic area, and risk factors. The most common causes of exudative pleural effusions have been shown to be primary pleural malignancy or metastasis and tuberculous pleuritis [1]. Due to the low yield of thoracocentesis and closed needle biopsy, 25% to 40% patients with pleural effusion remain undiagnosed [2,3]. Thoracoscopy overcomes the obstacles preventing such diagnosis and provides direct visualization and sampling of pleural cavity, thereby markedly improving the diagnostic yield. It is now considered to be one of the important endoscopic techniques in respiratory medicine. Compared to “surgical” thoracoscopy or video-assisted thoracic surgery (VATS), both of which are performed under general anaesthesia, medical thoracoscopy can be performed in an endoscopy suite under local anaesthesia or conscious sedation. There are various diagnostic and therapeutic indications for medical thoracoscopy like staging of lung cancer with pleural effusion, staging of pneumothorax, localized chest wall lesions, and talc poudrage in malignant pleural effusions and in pneumothorax. However, undiagnosed pleural effusion remains the most common indication by which the procedure is performed [4].
In recent years, medical thoracoscopy has come to well established as an important procedure that plays a crucial role in undiagnosed pleural effusion; however, instrument availability leads to its underutilization. Therefore, in this study, we have retrospectively analysed the outcome of single port rigid thoracoscopy in patients with undiagnosed pleural effusions.
Materials and Methods
1. Study plan
The current study included all patients who presented to our centre with undiagnosed pleural effusion between 2016 to 2020 and who underwent rigid medical thoracoscopy as a diagnostic procedure. Undiagnosed pleural effusion was defined as failure to achieve a diagnosis through initial pleural fluid analysis, including biochemical analysis, adenosine deaminase levels (ADA), culture and sensitivity, gram and Ziehl-Neelsen staining, and the existence of at least three pleural fluid samples with inconclusive cytology. However, among patients in which the pleural effusion showed atypical cells, those in which the cytopathological type for malignancy was not confirmed were also labelled as undiagnosed pleural effusion.
2. Methodology
All patients had undergone a routine clinical assessment, blood investigations, electrocardiograph, chest radiography, and thoracocentesis. The included patients had no obvious parenchymal lesion suggesting a specific diagnosis. The data collected included information on the patient’s demographics, clinical characteristics, primary thoracocentesis profile including pleural fluid cytology, pleural fluid protein, glucose, ADA, and differential count, thoracoscopy findings, biopsy report, and post procedure complications. Pleural effusion was diagnosed as exudative using Light’s criteria. Patients with ADA <40 U/L were labelled as low ADA [5].
3. Rigid thoracoscopy
Thoracoscopy was performed using a single port rigid thoracoscope in the endoscopy suite while the patient was under conscious sedation and local anaesthesia. Patient blood pressure, pulse rate, and oxygen saturation were continuously monitored. Supplementary oxygen was provided to maintain the oxygen saturation above 90%. The patient was positioned in the lateral decubitus position, with the unaffected lung in the dependent position and the arm raised above the head. The involved hemithorax was disinfected, and 15 to 30 mL of lidocaine 2% was injected at the point of entry, through all layers of chest wall until the pleura. A single puncture, which involved a 1.5 to 2 cm incision in the mid-axillary line between the fourth and seventh intercostal spaces of the chest wall, was made, and a track was created through blunt dissection. Next, a trocar was introduced, and the pleural cavity was punctured. Pleural fluid was aspirated using a small-diameter catheter, after which the pleural cavity was fully examined. Biopsy specimens (4 to 5 in number) of parietal pleura were taken as appropriate under direct vision. Finally, a chest tube was inserted, and lung expansion was radiographically confirmed before removal of the tube [5]. A chest radiography was taken within 24 hours. The obtained specimens obtained were preserved in 10% neutral buffered formalin and normal saline for microbiological examination. Then, sections were examined by the pathologist who also knew the clinical data. A final diagnosis was then made considering the biopsy findings regarding both histopathological and microbiological examinations, and further investigations were conducted according to individual patient circumstances and the subsequent clinical course.
4. Ethical approval
This study was reviewed and approved by the Institutional Ethics Committee (IEC) of the Shri Guru Ram Rai institute of Medical and Health Sciences, Dehradun, Uttrakhand, India (IEC approval number: SGRR/IEC/04/23). Written informed consent was waived off due to retrospective nature of study. The study was performed in accordance with the principles of the Declaration of Helsinki.
Results
In total, 92 patients underwent rigid medical thoracoscopy as a diagnostic procedure for undiagnosed pleural effusion in our centre; as eight patients were excluded due to non-availability of data, the data of 84 patients was ultimately analysed. Of these 84 patients, 56 (67%) were male and 28 (33%) were female. Among all patients, the most common symptom was shortness of breath, followed by chest pain and cough; most patients had multiple respiratory symptoms. Table 1 lists the demographics, clinical characteristics, and primary thoracocentesis profile of the patients. Cytological samples showed lymphocytic predominance in 82 patients, and two patients had neutrophilic predominance: These two patients did not improve with antibiotics and no pathogenic organism could be isolated on culture. Out of 82 lymphocytic exudative pleural effusions, 22 patients had atypical cells on pleural fluid cytology; however, the cytopathological type for malignancy was not confirmed for these patients. In 52.9% of the cases showing yellow coloured pleural fluid on aspiration, histopathology was suggestive of malignant effusion; however, 86% of hemorrhagic effusions were malignant.
Table 2 presents the findings of gross examination of pleura on thoracoscopy and histopathological diagnosis. Figure 1A, B shows the thoracoscopic appearance and histopathological appearance of the pleural lesions. Rigid thoracoscope used in this study is shown in Figure 1C. Pleural nodules (63%) were the most common finding, and malignancy was common diagnosis in pleural nodules. Three to four biopsy samples were taken in each patient, and the average biopsy sample size was 18 mm.
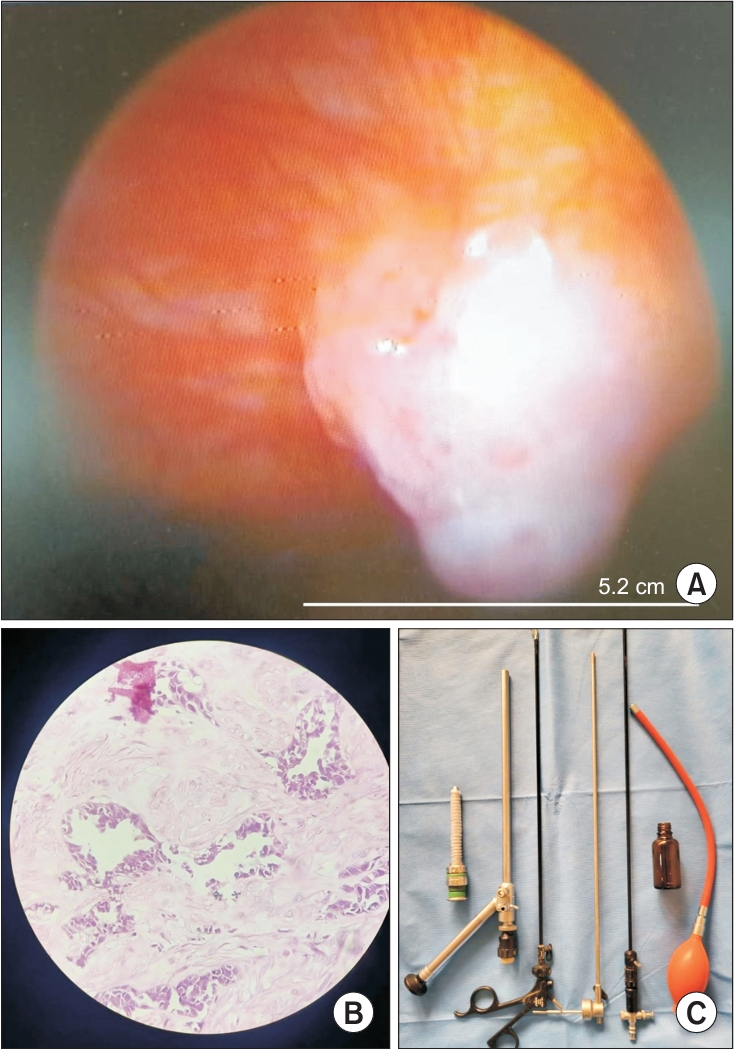
(A) Thoracoscopic view of pleural based mass. (B) H&E-stained section of pleural biopsy showing tumour cells arranged in glands (40× magnification with high power view). (C) Single puncture rigid thoracoscope with trocar and biopsy forceps.
Malignant pleural effusion was diagnosed in 61 (72.6%) patients, with the lung adenocarcinoma being the most common histopathological subtype, appearing in 36 (42.6%) patients. Tuberculosis was reported in 18 (21.4%) patients. Poorly differentiated carcinoma was another common subtype, and other reported malignancies were mesothelioma, small cell carcinoma, squamous cell carcinoma, and metastasis from breast and ovary tumours (Table 3). Overall, four patients had non-specific chronic inflammation in biopsy report, and only one patient had an inconclusive report. These five patients were kept under observation for 12 weeks and pleural effusion was found to resolved gradually, thus suggesting a benign aetiology of pleural effusion. No major complications were reported in any of the patients, but four patients developed minor complications: Two patients had persistent air leak and two patients developed subcutaneous emphysema which resolved with conservative management.
Discussion
Pleural effusion can be a presenting feature of numerous diseases, and it is sometimes the only clinical presentation of a disease. Thoracentesis and closed needle biopsy are the diagnostic procedures that are initially used for adequate diagnosis in suitable patients. However, the yield of closed needle biopsy in diagnosing tubercular pleuritis is only 33% [6], and it is even lower in malignant diseases. One study showed a 17% yield for closed needle biopsy in pleural malignancy [3].
Medical thoracoscopy has been developed as a newer intervention in the field of pulmonology. It is a minimally invasive procedure that can be performed by a pulmonary physician alone while the patient is under local anaesthesia. Previous studies have shown that medical thoracoscopy achieves remarkable results in undiagnosed pleural effusions [7]. The present study has reviewed the data of 84 patients with pleural effusion who remained undiagnosed after a basic work-up. Thoracocentesis revealed atypical cell in 26 patients, but histopathological diagnosis was not confirmed, thus necessitating medical thoracoscopy. Two types of thoracoscopes are available for thoracoscopy: a rigid thoracoscope and a semi-rigid thoracoscope, also known as a pleuroscope. Although studies have shown that rigid thoracoscopy can obtain samples with a higher diagnostic value than semi-rigid thoracoscopy (specifically regarding mesothelioma diagnosis) [8,9], there is still a general lack of ease and familiarity with rigid thoracoscopy among practitioners. In our study, 79 (94%) patients were given confirmed diagnostic results, thus indicating an impressive yield that is consistent with those reported in previous studies [8-10].
The presence of macroscopic findings on thoracoscopy increases the probability of histopathological diagnosis, and in our study, pleural nodule was found to be the most common macroscopic findings, as malignancy was reported in 83% of pleural nodules. The pleural mass clearly depicts the malignant pathology, and all patients in our study who were found to have pleural mass on direct visualization were malignant. The average biopsy size in our study was 18 mm, which might explain the high yield of rigid thoracoscopy. Similarly, in a study by Dhooria et al. [10], the biopsy size of rigid thoracoscopy was comparable to that in our study as well as much higher than that of a biopsy obtained by a semi-flexible thoracoscope. Similar to the findings of prior studies, the most common histopathological diagnosis in the current study was malignancy, with adenocarcinoma being the most common subtype [11,12], followed by tuberculosis.
Regarding the safety of rigid thoracoscopy, our study showed good results, as there were no major complications in any patients. Only minor complications like subcutaneous emphysema and air leaks were seen in four patients; such minor complications have also been commonly seen in previous studies [10,12,13].
Overall, rigid thoracoscopy has the advantages of a larger biopsy size and a higher diagnostic yield with a similar complication rate. However, the yield was similar when a successful biopsy was taken [10,14]. An operator in one trial also rated the ease of taking a biopsy sample as significantly higher than the corresponding ease of doing so with the rigid instrument. The reported disadvantages included the slightly larger scar size and the fact that the image quality was better with the semi-rigid instrument [14]. Prior studies have compared the cost of semi-rigid thoracoscopy versus that of VATS, and a previous review showed that the procedure-related cost for patients that received semi-rigid thoracoscopy was significantly lower than that for patients who received VATS [15]. No study has compared the cost of semi-rigid versus single port rigid thoracoscopy, but the much lower cost of the instrument for the latter strengthens the potential utility of rigid thoracoscopy in undiagnosed pleural effusion, particularly in developing countries like India, where cost is a major roadblock in instrument procurement. Single port rigid thoracoscopy is a safe and well-tolerated procedure that yields a bigger biopsy with high diagnostic yield. It is clearly important for undiagnosed pleural effusion to be subjected to medical thoracoscopy. The lower cost of the rigid instrument may lead to wider availability and hence ultimately improved diagnostics.
Notes
Authors’ Contributions
Conceptualization: Rawat J, Jangpangi DS. Methodology: Kumar A, Mrigpuri P. Formal analysis: Singh AP, Bhatt R. Data curation: Kumar A, Mrigpuri P. Software: Kumar A, Mrigpuri P, Bhatt R. Validation: Rawat J, Jangpangi DS, Mrigpuri P. Investigation: Kumar A, Mrigpuri P, Singh AP. Writing - original draft preparation: Rawat J, Kumar A, Mrigpuri P. Writing - review and editing: Kumar A, Mrigpuri P, Bhatt R. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
No funding to declare.
Acknowledgements
The authors are indebted to their colleagues who referred patients, in particular Dr. Tejas Sood and Dr Aabid Shafi Wani. for helping on technical aspects. We are thankful to Mr. Sanjeev Kumar, Technician, Intervention Suite for helping in study right from registration and technical aspects in performing thoracoscopy.