Recent Advances in Adjuvant Therapy for Non–Small-Cell Lung Cancer
Article information
Abstract
After the successful development of targeted therapy and immunotherapy for the treatment of advanced-stage non-small cell lung cancer (NSCLC), these innovative treatment options are rapidly being applied in the adjuvant setting for early-stage NSCLC. Some adjuvants that have recently been approved include osimertinib for epidermal growth factor receptor-mutated tumors and atezolizumab and pembrolizumab for selected patients with resectable NSCLC. Numerous studies on various targeted therapies and immunotherapy with or without chemotherapy are currently ongoing in the adjuvant setting. However, several questions regarding optimal strategies for adjuvant treatment remain unanswered. The present review summarizes the available literature, focusing on recent advances and ongoing trials with targeted therapy and immunotherapy in the adjuvant treatment of early-stage NSCLC.
Introduction
In 2020, lung cancer emerged as the most common type of cancer in Korea, excluding thyroid cancer [1]. Furthermore, it is predicted that lung cancer will become the most common type and cause of cancer-related deaths in Korea by 2023 [2].
Regarding stage distribution at diagnosis, lung cancer accounts for the highest proportion of patients diagnosed at a distant metastatic stage in Korea [3]. However, in the past few decades, the demographics of patients with lung cancer have changed, with an increasing number of patients diagnosed at localized stages (20.0% men and 58.7% women) [4]. This is mainly due to decreased smoking prevalence, improved staging with 18F-fluorodeoxyglucose-positron emission tomography and magnetic resonance imaging of the brain, and early screening programs using low-dose chest computed tomography [4-6]. Therefore, the importance of therapeutic interventions to cure early-stage lung cancer, in addition to surgical treatment, is gradually increasing as its incidence rises.
The traditional standard of care for patients with early-stage lung cancer is surgical resection followed by adjuvant cytotoxic chemotherapy in pathological stages II–III and high-risk stage IB (including poorly differentiated tumors, vascular invasion, wedge resection, visceral pleural involvement, and unknown lymph node status) [7]. Recently, two randomized control trials (RCTs), which prompted changes in clinical practice, were published in 2020 [8] and 2021 [9]. In the present review, we briefly discuss recent advances in the adjuvant treatment of non-small cell lung cancer (NSCLC).
Cytotoxic Chemotherapy
According to the Korean Association of Lung Cancer Registry, the 5-year relative survival rates of patients with stage I, II, III, and IV NSCLC are 82%, 59%, 16%, and 10%, respectively [10]. However, patients who undergo surgery remain at a substantial risk of recurrence even after complete disease resection. Indeed, it is estimated that 30% to 75% of patients with NSCLC who undergo surgery with curative intent develop recurrence and eventually die of their disease after 8 to 14 months [11].
Cytotoxic chemotherapy remains the standard adjuvant treatment for early-stage NSCLC. In addition to successful surgery, adjuvant cisplatin-based chemotherapy has demonstrated a survival advantage, especially in patients with stage II and III disease. Lung Adjuvant Cisplatin Evaluation, a pooled analysis of patient data from the five largest trials of cisplatin-based che-motherapy for completely resected stage I–III NSCLC, demonstrated a disease-free survival (DFS) benefit of 5.8% and 5-year absolute survival benefit of 5.4%. However, subsets showed possible harm in patients with stage IA and performance status 2, mainly because of chemotherapy toxicity [12]. As Cancer and Leukemia Group B 9633 (CALBG9633), an RCT designed to study adjuvant paclitaxel/carboplatin in stage IB NSCLC, and the the North American Intergroup phase III trial of adjuvant cisplatin plus vinorelbine (JBR-10) study, the North American Intergroup Study of adjuvant cisplatin and vinorelbine, demonstrated survival benefit of adjuvant therapy in patients with tumors ≥4 cm, the population considered most appropriate for adjuvant therapy is now stage IIA (T2bN0) or greater [13,14].
Targeted Therapy
As adjuvant-targeted treatments, gefitinib, icotinib, erlotinib, and osimertinib showed greater benefits than cytotoxic chemotherapy in patients with epidermal growth factor receptor (EGFR) mutations. Among them, osimertinib was the first approved targeted therapy for EGFR-mutated NSCLC in an adjuvant setting [15].
The Gefitinib Versus Vinorelbine/Platinum as Adjuvant Treatment in Stage II-IIIA (N1-N2) NSCLC With EGFR Mutation (ADJUVANT)-Chinese Thoracic Oncology Group 1104 (CTONG1104) [16] and adjuvant gefitinib versus cisplatin and vinorelbine in completely resected (stage II-III) NSCLC patients with mutated EGFR (IMPACT) [17] trials are two representative phase III trials that reported 5-year overall survival (OS) results of adjuvant gefitinib. The ADJUVANT-CTONG1104 trial first reported positive DFS for adjuvant treatment with gefitinib, compared with cisplatin and vinorelbine chemotherapy, in patients with resected stage II–IIIA EGFR-mutated NSCLC in 2017 in China. The median DFS was significantly longer in the gefitinib arm (30.8 months vs. 19.8 months: hazard ratio [HR], 0.56; 95% confidence interval [CI], 0.40 to 0.79), with a DFS rate of 39.6% vs. 32.5% and 22.6% vs. 23.2% at 3 and 5 years, respectively. Nevertheless, the benefit in DFS did not translate into survival, with a statistically insignificant OS advantage (median 75.5 months vs. 62.8 months: HR, 0.92; 95% CI, 0.62 to 1.36) [16]. Since then, several trials have presented similar DFS benefit with various first-generation tyrosine kinase inhibitors (TKIs), such as erlotinib (erlotinib versus vinorelbine plus cisplatin as adjuvant therapy in Chinese patients with stage IIIA EGFR-mutant NSCLC [EVAN], 2018 [18]; adjuvant erlotinib in patients with resected EGFR-mutant NSCLC [SELECT], 2019 [19]) and icotinib (icotinib versus chemotherapy as adjuvant treatment for stage II–IIIA EGFR-mutant NSCLC [EVIDENCE], 2021 [20]).
In Japan, a similarly designed IMPACT trial failed to demonstrate the superiority of gefitinib in terms of DFS and OS end points [17]. The IMPACT study was an open-label, randomized, phase III trial that evaluated the efficacy of adjuvant gefitinib versus cisplatin and vinorelbine chemotherapy in patients with resected stage II–III EGFR-mutated NSCLC. The median DFS was 35.9 and 25.1 months in the gefitinib and cisplatin/vinorelbine groups, respectively. However, the Kaplan-Meier curves crossed around 4 years after surgery, with no statistically significant difference (HR, 0.92; 95% CI, 0.67 to 1.28). OS was also not different (HR, 1.03; 95% CI, 0.65 to 1.65), with the 5-year OS rates being 78.0% and 74.6% in the gefitinib and cisplatin/vinorelbine groups, respectively [17]. This negative evidence for first-generation adjuvant TKI highlights the importance of drug choice and therapy duration in this setting [21].
The ADAURA study is a phase III placebo-controlled trial evaluating adjuvant osimertinib in patients with stage IB to IIIA EGFR-mutated resected NSCLC for 3 years. In the 2020 ADAURA primary analysis, the HR of DFS of the primary efficacy population (stage II to IIIA NSCLC) was 0.17 (95% CI, 0.12 to 0.23) [8]. Based on these data, the U.S. Food and Drug Administration (FDA) approved osimertinib as an adjuvant-targeted therapy for EGFR-mutated NSCLC, regardless of adjuvant chemotherapy, on December 18, 2020 [15]. Updated data after 2 additional years of follow-up showed a sustained DFS benefit with osimertinib (HR, 0.23; 95% CI, 0.18 to 0.30) [22] . In addition, adjuvant osimertinib reduced the risk of central nervous system recurrence (HR for central nervous system disease recurrence or death among patients with stage IB to IIIA disease, 0.18 in the primary analysis and 0.36 in the updated analysis) [8,22]. The median follow-up duration for OS in all patients with stage II–IIIA disease was 59.9 months in the osimertinib group and 56.2 months in the placebo group. The 5-year OS was 85% (95% CI, 79 to 89) in the osimertinib group and 73% (95% CI, 66 to 78) in the placebo group (HR, 0.49; 95% CI, 0.33 to 0.73) [23]. A comparison of the efficacy outcomes of recent adjuvant EGFR TKI trials is shown in Table 1.
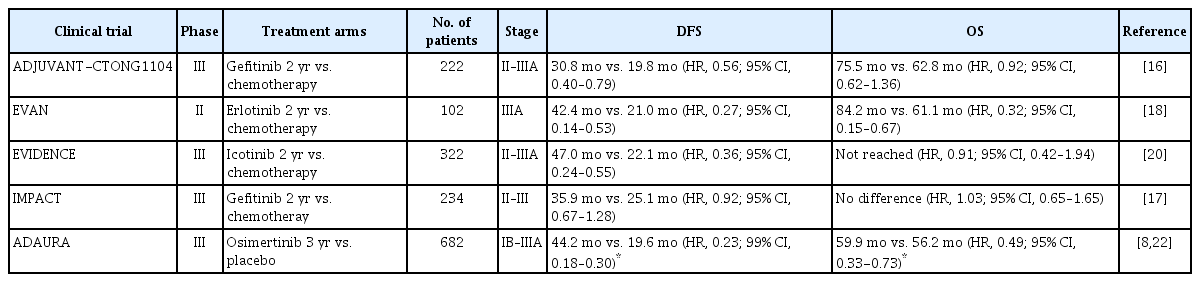
Comparison of clinical trials involving adjuvant epidermal growth factor receptor tyrosine kinase inhibitors
Despite the promising results of adjuvant EGFR TKIs, several controversies remain. The first question concerns the necessity of adjuvant chemotherapy. The CTONG1104, EVIDENCE, and IMPACT studies were head-to-head comparisons between EGFR TKIs and chemotherapy, which are different from ADAURA (osimertinib versus placebo with or without adjuvant chemotherapy). Wu et al. [24] reported that there was no apparent significant difference in the DFS impact of osimertinib among those who did (HR, 0.16; 95% CI, 0.1 to 0.26) or did not (HR, 0.23; 95% CI, 0.13 to 0.4) receive adjuvant chemotherapy in the ADAURA study. Moreover, the ADAURA study recently demonstrated a significant OS benefit with the adjuvant osimertinib [23]. To date, adjuvant chemotherapy remains recommended for resected stage II–IIIA NSCLC before the use of adjuvant osimertinib. However, future studies should address this question.
Another issue concerns the duration of treatment. The treatment duration of osimertinib was 3 years in the ADAURA trial and 2 years for first-generation TKIs in the CTONG1104, EVAN, and IMPACT trials. The advantage of first-generation TKIs persisted during the treatment period and the survival benefit lasted for almost 2 years after treatment cessation. Therefore, the optimal duration of adjuvant TKI therapy remains unclear and requires further investigation. The detection of minimal residual disease has been suggested as a potential treatment decision approach [23]. In addition, the ongoing 5 years of adjuvant osimertinib treatment (phase 2 single-group A Study of 5 Years of Adjuvant Osimertinib in Completely Resected Epidermal Growth Factor Receptor Mutation [EGFRm] NSCLC [TARGET]; NCT05526755) is anticipated to resolve this issue. These and other unanswered questions may be addressed in future trials.
In addition to EGFR mutations, other oncogenic driver mutations, including anaplastic lymphoma kinase (ALK), ROS proto-oncogene 1 (ROS1), neurotrophic tyrosine kinase receptor (NTRK), rearranged during transfection proto-oncogene (RET), and v-Raf murine sarcoma viral oncogene homolog B1 (BRAF), have been investigated. However, compared to EGFR-mutated NSCLC, there is a lack of large, randomized trial outcomes in early-stage ALK-positive NSCLC [25,26]. Therefore, ALK TKIs are not yet FDA-approved in the adjuvant setting.
There are several active phase III trials for adjuvant ALK TKIs. Firstly, Adjuvant Lung Cancer Enrichment Marker Identification and Sequencing Trial (ALCHEMIST)-ALK (NCT02201992) is a randomized, phase III trial evaluating adjuvant crizotinib in patients with stage IB–IIIA ALK-positive resected NSCLC for 2 years, with a primary endpoint of OS [7,27]. The actual start of the study was in 2015 and it is estimated to be completed by 2036 [7]. Secondly, the A Study Comparing Adjuvant Alectinib Versus Adjuvant Platinum-Based Chemotherapy in Patients With ALK Positive Non-Small Cell Lung Cancer (ALINA) trial is a randomized, phase III study designed to investigate the efficacy and safety of alectinib, compared with platinum-based chemotherapy, in the adjuvant setting for 2 years, with a primary endpoint of DFS [28,29]. This trial opened in 2018 with an estimated study completion in 2026 [29]. Lastly, another second-generation ALK TKI, ensartinib, is being evaluated as an adjuvant treatment in ALK-positive stage II–IIIB NSCLC after surgical resection with or without chemotherapy (NCT05341583) [30]. In summary, several adjuvant ALK TKIs trials are currently underway; however, due to delayed patient recruitment, it is anticipated that the results of these trials will only be available in several years [25,26]. Table 2 shows the details of ongoing phase III RCTs of adjuvant-targeted therapy [7,29-36].
Immunotherapy
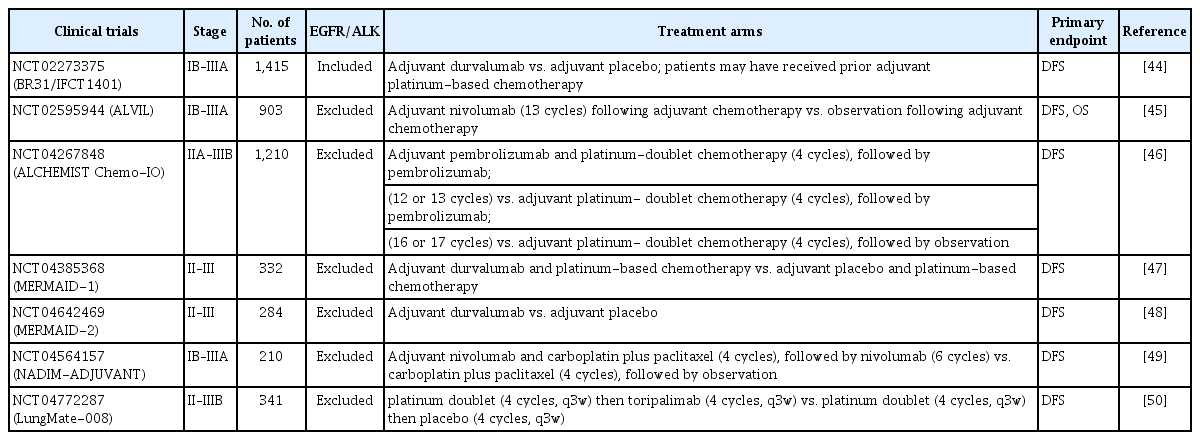
Immune checkpoint inhibitors (ICIs) have revolutionized advanced NSCLC treatment. Based on the success of ICIs in advanced settings, there is growing interest in expanding their use in patients with early-stage disease. The Study to Assess Safety and Efficacy of Atezolizumab (MPDL3280A) Compared to Best Supportive Care Following Chemotherapy in Patients With Lung Cancer (IMpower010) [9] and Study of Pembrolizumab (MK-3475) vs Placebo for Participants With Non-small Cell Lung Cancer After Resection With or Without Standard Adjuvant Therapy (MK-3475-091/KEYNOTE-091) (PEARLS) trials [37] have published results that suggest the effectiveness of ICIs in the adjuvant setting.
The IMpower010 trial was the first phase III randomized study to show a significant improvement in DFS with ICIs following adjuvant cisplatin-based chemotherapy in patients with resected early-stage NSCLC [9]. IMpower010 is an open-label phase III study with a sample size of 1,280 patients with resected stage IB–IIIA NSCLC. After completing up to four cycles of adjuvant cisplatin-based chemotherapy, eligible participants were randomized to receive 16 cycles of atezolizumab or best supportive care. A DFS benefit was observed in the experimental arm of stage II–IIIA patients with programmed death-ligand 1 (PD-L1) ≥1% (HR, 0.66; 95% CI, 0.50 to 0.88). It was even higher in patients with PD-L1 ≥50% (HR, 0.43; 95% CI, 0.27 to 0.68). No clear benefits were observed in the EGFR or ALK subgroups [9]. OS data are still immature; however, the HR for OS at this early timepoint was 0.99 (95% CI, 0.73 to 1.33) among all patients with stage II–IIIA disease and 0.77 (95% CI, 0.51 to 1.17) in the subgroup of patients with PD-L1-positive tumors [38]. Furthermore, the OS interim analysis results were presented at the World Conference on Lung Cancer in 2022. After a median follow-up of 45.3 months, the atezolizumab group showed a better OS trend than the best supportive care group among patients with PD-L1 ≥1% with stage II–IIIA NSCLC (5-year OS rate: 76.8% vs. 67.5%). For patients with PD-L1 ≥50% stage II–IIIA NSCLC without EGFR mutations or ALK rearrangements, the 5-year OS rates in the atezolizumab and control groups were 84.8% and 67.5%, respectively (HR, 0.42; 95% CI, 0.23 to 0.78) [39]. Based on the findings of this trial, adjuvant atezolizumab was approved by the FDA for use in patients with completely resected stage II–IIIA NSCLC with PD-L1 expression on ≥1% of tumor cells [40]. However, there is a discrepancy between the cut-off level of PD-L1 expression in FDA and Korea FDA approvals for adjuvant atezolizumab (FDA: PD-L1 ≥1% vs. Korea FDA: ≥50%) [41].
The PEARLS/KEYNOTE-091 trial is another key phase III randomized trial of adjuvant immunotherapy. The PEARLS/KEYNOTE-091 trial is a triple-blind phase III trial with a sample size of 1,177 patients with completely resected, PD-L1-positive stage IB–IIIA NSCLC. Adjuvant chemotherapy was not mandatory but was to be considered for patients with stage IB disease and strongly recommended for those with stage II and IIIA disease. Adjuvant pembrolizumab improved DFS compared with that obtained with the placebo (54 months vs. 42 months: HR, 0.76; 95% CI, 0.63 to 0.91), with a nonsignificant trend toward improvement in those with tumor expression of PD-L1 ≥50% (median DFS not reached in either arm: HR, 0.82; 95% CI, 0.57 to 1.18). The significance boundary for OS in the intention-to-treat population was not crossed (18-month rate, 91.7% vs. 91.3%); however, the results were immature [37]. On January 26, 2023, the FDA approved pembrolizumab for adjuvant treatment following resection and platinum-based chemotherapy for stage IB (T2a ≥4 cm), II, or IIIA NSCLC [42]. However, adjuvant pembrolizumab has not yet received regulatory approval in South Korea.
Similar to targeted therapy, adjuvant immunotherapy is riddled with several challenges, such as predictive biomarkers for the benefit of ICIs, efficacy for oncogene-mutated NSCLC, timing of ICIs, combined regimen, duration of treatment, and selection between neoadjuvant and adjuvant purposes [43]. Ongoing and future trials will address these unmet requirements. Table 3 summarizes phase III randomized trials that have investigated adjuvant immunotherapy [44-50].
Conclusion
Although the survival benefits of adjuvant chemotherapy are statistically significant, there is an urgent need for enhanced treatment strategies to further improve patient survival. Adjuvant-targeted therapy and immunotherapy may improve the outcomes of selected patients, and ongoing studies may further improve survival. To date, osimertinib for EGFR-mutated NSCLC and ICIs, such as atezolizumab and pembrolizumab, have received FDA approval for adjuvant NSCLC therapy. Ongoing clinical trials and mature data from completed trials will help shed light on the remaining doubts and controversies. These advances will improve the cure rates of early-stage resectable NSCLC.
Notes
Authors’ Contributions
Conceptualization: Kim MH, Lee MK, Eom JS. Writing - original draft preparation: Kim MH, Kim SH. Writing - review and editing: Kim MH, Eom JS. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
The present study was supported by a clinical research grant from the Pusan National University Hospital (2023). This funding source had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.