Comparing Inhaler Use Technique Based on Inhaler Type in Elderly Patients with Respiratory Disease
Article information
Abstract
Background
The aim of this study was to investigate inhaler device handling in elderly patients. Inhaler devices with respect to misuse and error correction were also compared.
Methods
Inhaler use technique was assessed using standardized checklists at the first visit and 3-month follow-up visit after retraining. The primary outcome was difference in the acceptable use ratio among inhaler devices. Secondary outcomes included differences in error correction, the most common step of misuse, and factors affecting the accuracy of inhaler use.
Results
A total of 251 patients (mean age, 76.4 years) were included. The handling of 320 devices was assessed in the study. All patients had been trained before. However, only 24.7% of them used inhalers correctly. Proportions of acceptable use for Evohaler, Respimat, Turbuhaler, Ellipta, and Breezhaler/Handihaler were 38.7%, 50.0%, 61.4%, 60.8%, and 43.2%, respectively (p=0.026). At the second visit, the acceptable use ratio had increased. There were no significant differences among inhaler types (Evohaler, 63.9%; Respimat, 86.1%; Turbuhaler, 74.3%; Ellipta, 64.6%; and Breezhaler/Handihaler, 65.3% [p=0.129]). In multivariate analysis, body mass index, Turbuhaler, and Ellipta showed positive correlations with acceptable use of inhalers, whereas Chronic Obstructive Pulmonary Disease Assessment Test score showed a negative correlation.
Conclusion
Although new inhalers have been developed, the accuracy of inhaler use remains low. Elderly patients showed more errors when using pressurized metered-dose inhalers than using dry powder inhalers and soft-mist inhalers. However, there were no significant differences in misuse among inhaler devices after individual training. Results of this study suggests that repeat training is more important than inhaler type.
Introduction
Inhaled therapy is the mainstay of treatment of airway diseases, such as asthma [1] chronic obstructive pulmonary disease (COPD) and tuberculosis-destroyed lung with airflow limitation [2,3], owing to the rapid onset of action and occurrence of fewer adverse events. Since the launch of inhalers in the 1960s, more than 250 inhaler devices have become available for effective treatment [4]. Following the introduction of pressurized metered-dose inhalers (pMDIs), several dry powder inhalers (DPI) and soft-mist inhalers were released, with differences in the spraying method, particle size, and device manipulation method [5]. However, the misuse of inhalers has been a longstanding discussion and is still a major concern for physicians [6-8]. Although decades have passed since the first inhaler was developed, the accuracy of inhaler use remains low at approximately 40% [8,9]. Poor inhaler technique is associated with poor disease control and frequent disease exacerbation [9,10].
The difficulty with correct inhaler use is more severe in elderly patients. Several studies revealed a significant positive correlation between the misuse of inhalers and age [11-13]. As cognitive function, physical strength and dexterity, memory, and executive planning deteriorate, errors in inhaler use increase in elderly patients.
Differences in the accuracy of inhaler use based on the inhaler type have been investigated in many studies. Two review studies were aimed at investigating whether the misuse differed with the type of inhaler or not but failed to elucidate which inhaler is more frequently misused [14,15]. However, some studies reported that the recently launched inhaler Ellipta had fewer errors of misuse compared to other inhalers [16,17].
The aim of this study was to evaluate how accurately inhalers are used by the elderly patients with respiratory disease and whether the usage accuracy varies with the inhaler type or not. In addition, we compared the inhaler devices with respect to error correction after training.
Materials and Methods
1. Study design
We conducted a prospective study to compare the accuracy of inhaler use among different inhaler types in elderly patients with respiratory disease at an outpatient clinic of the Veterans Health Service Medical Center, Seoul, South Korea, from June 2018 to July 2019. The accuracy of inhaler use was checked by a research investigator. If the patient made errors, the investigator demonstrated the correct inhaler use technique through a face-to-face discussion. Three trained research investigators checked the accuracy of the inhaler use technique with standardized checklists. We used a teach-to-goal approach to correct identified errors, giving feedback to the patient. We repeated this coaching process as many times as needed in order for patients to use inhaler correctly. After education, the accuracy of inhaler use was re-checked at the 3-month follow-up visit.
The study protocol was approved by the institutional review board of the Veterans Health Service Medical Center, Seoul (2018-04-035-003). This study complied with the principles laid down in the Declaration of Helsinki. Written informed consent was obtained from each patient before enrollment in the study.
2. Participants
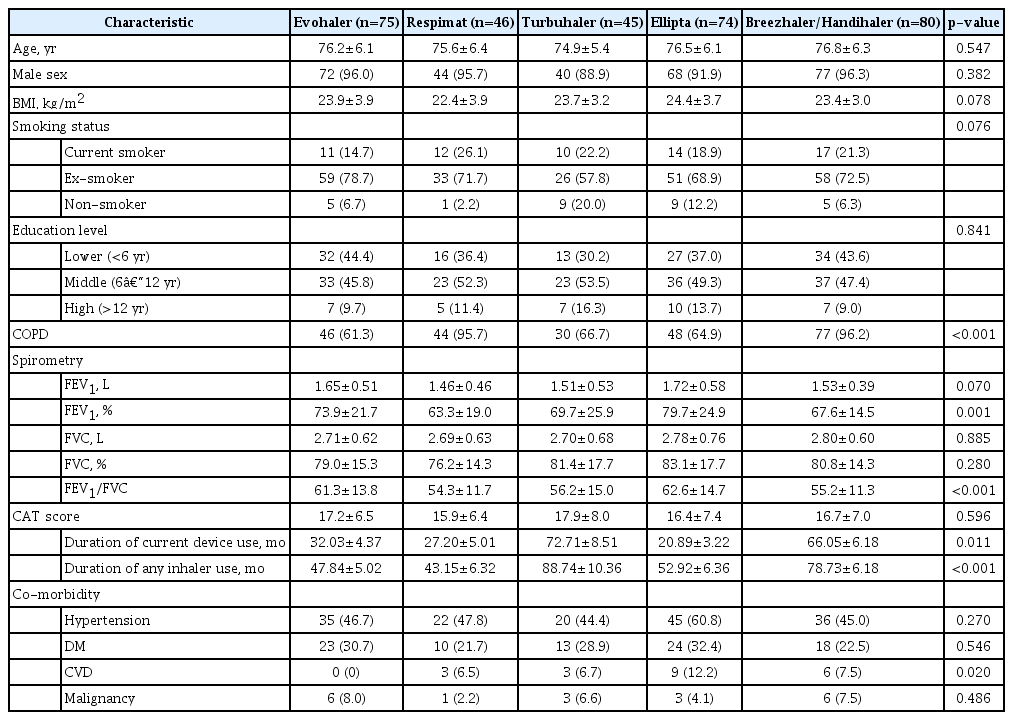
Patients with asthma or COPD, aged 65 years or older, who had been using an inhaler for at least 3 months in addition to receiving inhaler training previously were enrolled in the study. Patients provided informed consent to participate in this study. We checked the inhaler use accuracy at the first and 3-month follow-up visits. COPD Assessment Test (CAT) scores, total duration of inhaler use, smoking history, education level, satisfaction, etc. were recorded. Spirometric analyses were performed at the time of inclusion in the study and 1 year later.
3. Checklists
We created a simple standardized checklist for this study. Regardless of the type of device, all checklists consisted of five steps. Steps 1 and 2 were to perform exhalation while assembling and holding the inhaler correctly. These steps varied from device to device. Step 3 was to breathing out fully. Step 4 was to perform deep inhalation. Step 5 was to hold the breath. Participants received a score of “0” for each incorrect step and a score of “1” for each correct step. The inhaler use was regarded “acceptable” if the patient performed four or more of the five steps correctly.
4. Outcomes
We compared five types of inhalers. The primary outcome was differences in the acceptable use ratio among inhaler devices. Secondary outcomes included differences in error correction after training, most common step of misuse, and factors affecting the accuracy of inhaler use. Further, we surveyed patient satisfaction with each device and found the most frequently error-prone steps. In this study, six inhaler device types were used by participants: Evohaler (GlaxoSmithKline, London, UK/Chiesi, Parma, Italy), Respimat (Boehringer Ingelheim, Ingelheim am Rhein, Germany), Turbuhaler (AstraZeneca, Lund, Sweden), Ellipta (GlaxoSmithKline), Breezhaler (Novartis, Basel, Switzerland), and Handihaler (Boehringer Ingelheim). However, Breezhaler and Handihaler were analyzed in the same group because of the similarities. Finally, five device types were compared.
5. Statistical analyses
Data on clinical characteristics are expressed as the mean±standard deviation or number (proportion). We used Fisher exact test and Kruskal-Wallis test, as appropriate, to compare baseline characteristics among groups. To identify the factors affecting acceptable use, we calculated the adjusted odds ratio and 95% confidence intervals using a logistic regression model. All analyses were performed using the STATA software, version 15 (StataCorp LP, College Station, TX, USA). Statistical significance was defined as a two-tailed p-value <0.05.
Results
A total of 251 patients were enrolled in the study. Table 1 summarized the baseline characteristics of the study participants. Patients included 234 men (93.2%), with a mean age of 76.38±0.39 years. The mean period of current inhaler device use was 37.60±2.83 months, and mean total period of inhaler use was 57.15±3.54 months. Two or more inhalers were used by 68 participants, so we assessed 320 devices in the study. Evohaler, Respimat, Turbuhaler, Ellipta, and Breezhaler/Handihaler were used by 75 (23.4%), 46 (14.4%), 45 (14.1%), 74 (23.1%), and 80 (25.0%) patients, respectively. At the first assessment, only 79 patients (24.7%) used the inhaler correctly.
1. Rate of acceptable users based on the inhaler device type
We defined “acceptable” if the patient performed four or more of the five steps in the checklist correctly. At the first assessment, there were 158 acceptable users (49.4%). There was a significant difference in the device use technique among the five types. Proportions of acceptable users for Evohaler, Respimat, Turbuhaler, Ellipta, and Breezhaler/Handihaler were 38.7%, 50.0%, 61.4%, 60.8%, and 43.2%, respectively (p=0.026) (Table 2, Figure 1). The mean checklist score (total of 5 points) was highest in Turbuhaler (3.71±1.16), followed by Ellipta (3.59±1.13), Respimat (3.46±1.38), Breezhaler/Handihaler (3.45±1.05), and Evohaler (2.89±1.54) (p=0.014) (Table 2).

Proportions of acceptable use at the first visit and the second visit after individual education. The acceptable use rate by device at the first visit was 37.3% for Evohaler, 47.8% for Respimat, 62.2% for Turbuhaler, 60.8% for Ellipta, and 43.8% for Breezhaler/Handihaler (p=0.026). At the second visit after one-to-one training, there was no difference in the number of acceptable users for each inhaler type: 40 (64.5%) for Evohaler, 31 (81.6%) for Respimat, 29 (76.3%) for Turbuhaler, 43 (66.2%) for Ellipta, and 49 (65.3%) for Breezhaler/Handihaler (p=0.280).
2. Rate of acceptable users based on the inhaler device type among patients aged over 75 years
We analyzed inhaler use in 119 patients aged over 75 years. Proportions of acceptable users for Evohaler, Respimat, Turbuhaler, Ellipta, and Breezhaler/Handihaler were 37.0%, 42.9%, 62.5%, 51.7%, and 51.5%, respectively (p=0.547). The accuracy of inhaler use based on the inhaler device type among patients aged over 75 years was similar to the overall results, but no statistical difference was found.
3. Differences in the acceptable user ratio of the inhaler devices after individual education
When errors were corrected through one-to-one training and re-evaluated 3 months later, there were no differences in the rate of acceptable users among inhaler device types. Acceptable users for Evohaler, Respimat, Turbuhaler, Ellipta, and Breezhaler/Handihaler were 40 (64.5%), 31 (81.6%), 29 (76.3%), 43 (66.2%), and 49 (65.3%), respectively (p=0.280) (Table 3, Figure 1). The mean checklist scores (total of 5 points) also increased in all groups after training, but there was no difference between devices (Turbuhaler [4.13±0.84], Respimat [4.11±1.03], Ellipta [3.86±0.97], Breezhaler/Handihaler [3.84±1.12], and Evohaler [3.79±1.19], p=0.423) (Table 3).
4. Multivariate analysis of factors affecting inhaler use
Table 4 shows the results of univariate and multivariate logistic regression analyses of factors associated with the acceptable inhaler use in the baseline assessment. In the univariate analysis, the inhaler type, CAT score, body mass index (BMI), and the educational level were significantly associated with the acceptable use. No significant association with inhaler use was observed for patient age, presence of comorbidities, or period of use. In the multivariate analysis, the CAT score showed a negative correlation with the acceptable inhaler use whereas BMI, Turbuhaler, and Ellipta showed a positive correlation.
5. Comparison of the correct use rates at each step (based on the inhaler device) at baseline and after training
We evaluated the rate of correct handling of inhaler devices at each step at baseline and after training. Table 5 shows the results at the first visit and at the 3-month follow-up after training. Regardless of the type of inhaler device or training, patients performed “breathing out fully” least correctly. At the first visit, the percentage of patients who correctly performed the assembling step (pre-inhalation) was 64%–100%, and the patients who used Evohaler had the most errors (Evohaler, 64.9%; Respimat, 91.3%; Turbuhaler, 71.7%; Ellipta, 100%; and Breezhaler/Handihaler, 100%; p<0.001). For the fourth step of evaluating effective inhalation, which meant both full inhalation and coordination between inhalation and actuation, patients with Evohaler had the most errors (48% for Evohaler, 60.9% for Respimat, 93.3% for Turbuhaler, 82.4% for Ellipta, and 82.5% for Breezhaler/Handihaler; p<0.001). At the second visit after fixing the errors individually, the overall error rate had improved. However, errors by Evohaler users were significantly high in the assembling and inhalation steps.
Discussion
This was a prospective study performed to evaluate inhaler device usage, including newly developed devices, among elderly patients in the real world. Only 79 patients (24.7%) were used inhalers correctly, regardless of the inhaler device type. In a systematic review of errors in inhaler use from 1975 to 2014, Sanchis et al. [8] reported that the overall prevalence of the correct technique was only 31% (28%–35%) and has not improved over the past 40 years. Although new devices have been developed, these data are consistent with previous studies and demonstrate that the accuracy of inhaler use remains low.
We expected elderly patients to use DPIs better than pMDI owing to fewer coordination problems. In our study, Evohaler showed the lowest rate of acceptable inhaler technique among the five device types, and Turbuhaler and Ellipta showed fewer errors than MDI, with a statistical significance (p=0.026). In contrast, Respimat was not significantly different from DPI or MDI. After education, the significant differences in error frequency among devices disappeared. However, the analysis of each step revealed that Evohaler users performed more errors than users of other devices in the assembly (preinhalation) and inhalation steps even after training. Notably, Ellipta showed the lowest correction rate and Respimat showed the highest correction rate after one-on-one face-to-face training. We guess there are two main reasons. One is the selection bias arising from the design of this study. As a real-world study, we kept the device patients were using without randomly assigning devices to patients. Because Ellipta is easy to operate, there might be more patients with reduced cognitive and manipulation skills. The other reason for the lack of improvement is related to the stage at which the error appears. In the group of Ellipta, there were more errors in the exhalation (step 3), inhalation (step 4), and breath-hold (step 5) processes than in the preparation of device. In this study, the error of preparation step was relatively easy to be corrected, whereas the error of step 3–5 is difficult to improve. We thought that Respimat would require more individualized training, and if well-educated, errors could be reduced to a level over DPIs.
Although several studies showed that older age is a risk factor for incorrect use [13], the association between age and error is controversial [18,19]. In this study including the elderly people aged 65 years or above, age did not affect the accuracy of inhaler use in the multivariate analysis. Additionally, there was no difference in inhaler misuse between patients aged 65–75 and those aged 75 years or above. Diseases such as stroke or other factors that affect cognitive and behavioral skills, rather than age, may have a more significant impact on inhaler use [20]. Based on our results, we suggest that elderly patients can use inhalers suitably through repeated training.
Inhaler misuse is reportedly related to the device type (pMDI), female sex, lower education level, and lower socioeconomic status [21,22]. It is generally accepted that pMDI is more error prone than DPI, but studies are inconsistent in the findings of comparisons of DPIs, making it difficult to determine that a particular device shows fewer errors [22-24].
Many studies showed that educational intervention improves the accuracy of inhaler use. Klijn et al. [25] performed a systemic review of all studies related to education of inhaler technique and reported improvement in inhaler use, at least in the short term, with an average follow-up of 5 months. The teaching method, duration of education, individual, or group had no significant impact on outcomes [25]. Additionally, there was no difference in most variables correlated with poor inhalation between patients using pMDIs and those using DPIs [26]. The effectiveness of education also reportedly decreases over time [27,28]. Thus, it is recommended to train patients in inhaler use frequently, instead of switching inhalers [25]. The inhaler technique is unstable, so training on inhaler device use should be repeated to maintain correct use [18].
In this study, the most error-prone step was the third step of breathing out fully (correct users ratio, 46% [42%–50%]). This is a necessary step in inhaler use, regardless of the device type. As reported in previous studies, sufficient training is required for this step as well.
We assessed the accuracy of inhaler use in elderly patients, including new devices, and evaluated the degree of improvement after training and factors affecting inhaler use errors. However, there were limitations to the study. First, the patients kept the inhaler devices they were using and had not been randomly assigned to them. Therefore, it can be considered that the reality was reflected as it was, but our data cannot define an “easy-to-use inhaler.” The doctor would have already considered several factors related to the patient when choosing an inhaler. Physicians consider the ability to handle the device (97.9%), together with coordination (96.9%), ability to learn (87.5%), and adherence to therapy (84.4%) when selecting an inhaler device, based on the characteristics of each patient [29]. Second, we did not evaluate the patient’s cognitive or motor skills. We generally replaced these by checking the degree of education and history of stroke. Third, we could not measure the inspiratory flow and subjectively assessed the appropriate inhalation.
In conclusion, new inhalers have been developed, but the accuracy of inhaler use remains low in the elderly patients. The use of pMDI was associated with more errors compared to that of DPI or soft-mist inhalers, but there were no differences in misuse among inhaler devices after individual training. This study provides evidence that elderly patients need repeated training to use inhalers properly.
Notes
Authors’ Contributions
Conceptualization: Lee BJ. Methodology: Lee HY, Lee BJ, Chung KB. Formal analysis: Lee HY. Data curation: Lee HY, Won HK, Park Y, Chung KB, Lim HJ, Ahn YM. Software: Song JH, Chung KB. Investigation: Lee HY, Lee BJ. Writing - original draft preparation: Lee HY. Writing - review and editing: Lee BJ, Song JH, Won HK, Park Y, Chung KB, Lim HJ, Ahn YM. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
This study was supported by grants from VHS Veterans Medical Research Institute.