Introduction
Glucocorticoids are the most potent and reliable of the available agents among the anti-inflammatory drugs, and have assumed a major role in the management of asthma
1. However, long-term use of inhaled corticosteroids, especially at higher doses, has been accompanied by concern about systemic complications as well as local adverse events
2. Systemic complications of high dose inhaled steroids include osteoporosis, adrenocortical suppression, skin thinning, cataract, glaucoma and others
3. Local adverse events include throat pain or sore throat, dysphonia, oral candidiasis, and others
4.
Local adverse events of inhaled steroids are reported to be approximately 5~70%
4, and few studies have been reported because most of symptoms are mild compared to systemic complications. But, if appropriate managements of local adverse events are not taken, compliance of patients with inhaled steroids will be decreased and this may influence treatment of diseases. So the efforts that minimize local adverse events are needed.
The mouth rinsing method is currently implemented to prevent local adverse events by cleansing mouth immediately after taking inhaled corticosteroids. The mouth rinsing method has been practiced based on experience, and few studies have been made regarding the method. Toogood et al.
5 clinically identified oral candidiasis in 13% of asthma patients who took high dose inhaled corticosteroids. Moreover, Selroos and Halme
6 verified that plasma cortisol concentrations were significantly lower (p=0.007) in patients with rinsing mouth than not rinsing mouth when high dose of budesonide (1,600 ┬Ąg) was inhaled using dry powder inhaler (DPI). But, despite the practice of the mouth rinsing method, local adverse events induced by inhaled corticosteroids still result constraints in the use of medication to patients. For this reason, this study was established a hypothesis that the remained dry powder will be effectively removed when the patients do the method of immediate food intake after using inhaled corticosteroids. And since the administration methods of oral medications have been mostly recognized in relation to meals, the effects of training is thought to be accepted more generally when the use of inhaler devices will be guided as well. Therefore, the authors conducted a pilot study in order to verify the effectiveness of the immediate diet method reducing the occurrence of local adverse reactions.
Discussion
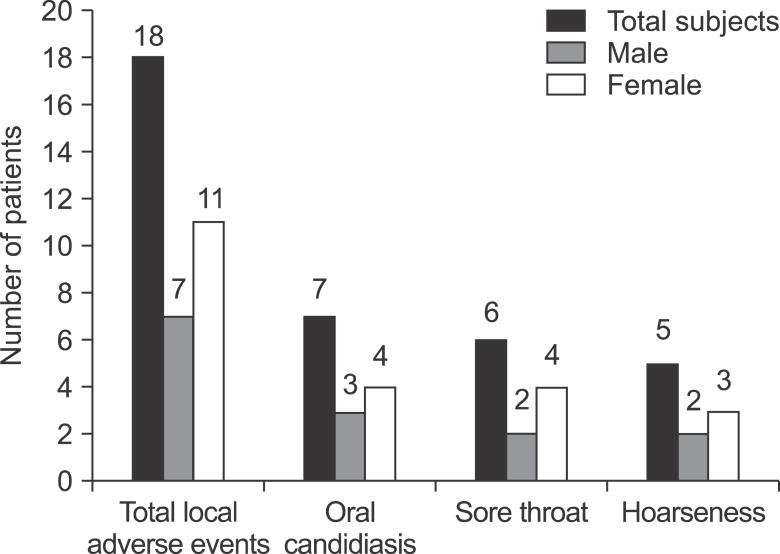
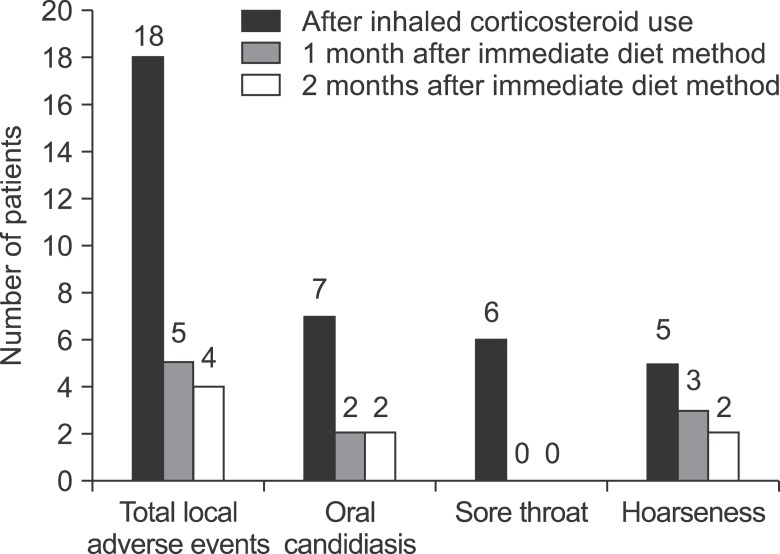
Among 98 subjects, 18 of them experienced local adverse events after taking inhaled corticosteroids. When the immediate diet method was instructed to those 18 patients, symptoms of 14 patients (77.8%) were relieved. Although mechanisms of improvement are unknown, the immediate diet method is thought to facilitate the elimination of dry power remaining in the mouth. And reducing the amount of steroid powder remaining in the mouth is known as a preventive measure for local adverse events. Yokoyama et al.
7 identified that the amount of steroid powder was reduced more than 90% when mouth rinsing was performed up to five times than when it was performed only once in patients who inhaled fluticasone. The results were demonstrated that local adverse events such as oral candidiasis are prevented when mouth rinsing is performed repeatedly to remove the remaining powder inducing adverse reactions. Similarly in our study, the reason for the reduced occurrence of local adverse events by using the immediate diet method was assumed that was due to removal of steroid powder in the mouth. When the improvement of each local adverse reaction was examined, six patients with sore throat were all improved after being trained with the immediate diet method. In case of patients with oral candidiasis, five out of seven got better after being trained with the immediate diet method. Moreover, three out of five patients with dysphonia showed improvement. The immediate diet method was suggested to be more effective especially in sore throat. However, further comparative studies are essential with larger study population to verify this prediction.
The incidence of local adverse events upon the use of inhaled corticosteroids vary from 5~70% depending on researchers
4,8,9. The occurrence of local adverse events differs depending on the ages of patients and types, container, and dose of inhalation devices. Dubus et al.
8 presented that 60% of pediatric patients reported local adverse events after taking inhaled corticosteroids. Pauwels et al.
10 reported that the incidence of cough, dysphonia, throat pain and other events were significantly increased when inhaled corticosteroids were taken using metered dose inhaler (MDI) instead of DPI (p=0.0001). According to meta-analysis in connection with this result, the risk of incidence was five times higher than placebo group in case of oral candidiasis when steroids were inhaled using the MDI. The risk of incidence was three times higher when DPI was used
11. Our study was comprised relatively older patients with the average age of 66. The prevalence rate of local adverse events was 18.4% when the patients took the drugs containing inhaled corticosteroids using DPI. Besides from oral candidiasis, dysphonia, and sore throat, other local adverse events include dermatitis around lips, cough, dry mouth, tongue hypertrophy, and others
4. In addition, there are some patients reporting toothache and stomatitis
12. However, in this study, there was no patient reporting symptoms other than oral candidiasis, sore throat, and dysphonia.
When mechanisms of those local adverse events were examined, immune functions of neutrophils, macrophages, and T-lymphocytes existing within the oral cavity were inhibited due to accumulation of inhaled corticosteroids in the mouth. This eventually leads to locally weakened immune system and increased glucose concentration in salivary glands, resulting rapid growth of
Candida species
13. Dysphonia is known to be resulted from deformation of vocal folds due to myopathy induced by accumulation of steroids in the muscles of vocal cord
14. Sore throat has been reported to be caused by secondary infection in the throat due to over-stimulated mucous membranes and weakened immune system
15. Therefore, when the patients with local adverse events perform the proper implementation of mouth rinsing or the immediate diet method, the amount of accumulated steroids within the oral cavity needs to be reduced. Besides, weakened local immune system in the throat will be enhanced. Moreover, secondary growth of
Candida species and throat infection will be prevented by reducing glucose concentration in salivary glands, leading to decreased adverse events including oral candidiasis, sore throat, and others. In addition, dysphonia induced by deformation of vocal cords is thought to be reduced by lowering accumulation of corticosteroids within the muscles.
Currently, mouth fresheners or gargles containing antifungal agents are prescribed for treatment in case of incidence of local adverse events such as oral candidiasis
16. When inhaled corticosteroids are used, patients are mostly instructed to practice mouth rinsing by immediately washing mouth with water to prevent the occurrence of local adverse events. In case of oral candidiasis, using MDI with spacer, or decrease in the frequency of inhaled corticosteroids are encouraged to reduce the amount of steroid powder remaining in the mouth. However, no clarification has been made on if up to what extent local adverse events could be reduced or prevented
6.
One of the limitations in this study is that the research was performed without control group even though it was a pilot study. To compare preventive effectiveness on local adverse events arising from inhaled corticosteroids, further studies are crucial with control groups in each of the mouth rinsing method currently applied based on previous experience and newly proposed the immediate diet method. Confirming possibilities of the preventive measures is essential for the progress of further studies. Second, no assessment or analysis has been made regarding on the difference varying depending on skillfulness of individual patient in the usage of inhalation device. However, this study was conducted on patients using the same container (diskus, DPI), and identical inhaled corticosteroids (fluticasone, 500 ┬Ąg/day) in the same dose. To minimize confusing factors, instructions with standardized methods were given by the trained nurse who was exclusively in charge of training patients regarding the use of inhalation device. Furthermore, according to a comparative study of Brand et al.
17 recently conducted on the MDI, accumulation of steroids within the oral cavity before and after the appropriate training of inhaler technique did not shown significant difference from 56% to 55%. According to a study of Lee and Yang
18, although knowledge and accuracy in the usage of inhalation device was increased with proper instructions conducted on patients, satisfaction on the use of inhaler was not increased and peak expiratory flow and instability in asthma control was not shown improvement either. Consequently, continuous trainings and elapsed time seemed to have almost no influence on the reduction of adverse reactions and the enhancement of pulmonary function. Additional studies are thought to be essential to investigate on the effectiveness of inhalation device training on reduction of steroids in the mouth and local adverse events. To progress further studies, analysis on the incidence of local adverse events needs to be performed collectively according to skillfulness assessed after training patients.
In summary, the immediate diet method is a method that having meals immediately after using inhaled corticosteroids to minimize local adverse reactions. This method is expected to be beneficial for reducing local adverse events of inhaled devices. So the results of this study will be used as basic data in upcoming comparative studies verifying effectiveness of the mouth rinsing method and the immediate diet method for reducing local adverse events.



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation






