Inhaled Corticosteroids Is Not Associated with the Risk of Pneumonia in Asthma
Article information
Abstract
The introduction of inhaled corticosteroids (ICS) for the management of asthma has led to a decrease in acute exacerbation of asthma. However, there are concerns regarding the safety of long-term ICS use, particularly pneumonia. Growing evidence indicates that ICS use is associated with an increased risk of pneumonia in patients with chronic obstructive pulmonary disease, whereas the risk in patients with asthma remains unclear. This review discusses the effect of ICS on pneumonia among patients with asthma to update the existing literature. Asthma is associated with an increased risk of pneumonia. Several hypotheses have been proposed to explain this association, including that asthma impairs the clearance of bacteria owing to chronic inflammation. Therefore, controlling airway inflammation with ICS may prevent the occurrence of pneumonia in asthma. In addition, two meta-analyses investigating randomized control trials showed that ICS use was associated with a protective effect against pneumonia in asthma.
Introduction
Asthma is a known risk factor for pneumonia. Talbot et al. showed an increasing incidence of invasive pneumococcal disease (IPD) in both children and adults (adjusted odds ratio [OR], 2.4; 95% confidence interval [CI], 1.9 to 3.1), whereas the OR for IPD was higher in patients with a high risk of asthma who experienced at least one or more hospitalizations or emergency room visits in the last year [1]. Shea et al. [2] further reported that compared with age-matched healthy controls, patients with asthma are more likely to have an approximately three-fold risk for all causes of pneumonia [1,2]. A systemic review of observational studies showed that asthma is a clear risk factor for community-acquired pneumonia (CAP) [3]. The occurrences of both uncomplicated pneumonia and pleural empyema increased in patients with asthma [4].
Asthma is a High Risk Factor for Pneumonia
The mechanism underlying this increased susceptibility to pneumonia in patients with asthma remains unclear, but several hypotheses have been proposed. First, patients with asthma have an increased prevalence of Streptococcus pneumoniae carriage during birth and childhood. The Copenhagen Prospective Study on Asthma in Childhood showed that infants colonized with S. pneumoniae had an increased risk of developing persistent wheezing or asthma [5]. Second, asthma impairs the clearance of pathogenic bacteria owing to increased production of mucin through hyperplasia of goblet cells, impaired antibody reactions, and airway obstructions [6,7]. Third, patients with asthma show alterations of the microbiome in the lungs and gut microbiota [8]. Controlling chronic airway inflammation and chronic bacterial infection is important to prevent pneumonia in asthma. Patients with asthma and poor adherence to inhaled corticosteroids (ICS) are known to be associated with uncontrolled airway inflammation and an increased risk of worsening asthma symptoms and exacerbations. Uncontrolled airway inflammation can lead to increased production of mucin and alteration in secreted mucus, resulting in abnormalities in viscosity and mucociliary clearance of the airway, which serves as a focus for localized infection, leading to invasive bacterial infections [6,9].
Several studies have suggested that ICS use could be the cause of higher susceptibility to pneumonia in patients with asthma. However, to validate this argument, randomized control trials (RCTs) or prospective studies must be conducted to show a higher prevalence of pneumonia among ICS users than among non-ICS users, with dose dependence. However, at present, this hypothesis is only supported by weak evidence from retrospective studies [10-13]. This association has yet to be demonstrated in RCTs, meta-analyses, and prospective studies [14-19]. This review summarizes the effect of ICS on pneumonia, focusing on the suggestion that ICS use is not associated with pneumonia in asthma.
Pneumonia Prevalence among ICS Users vs. Non-ICS Users with Asthma
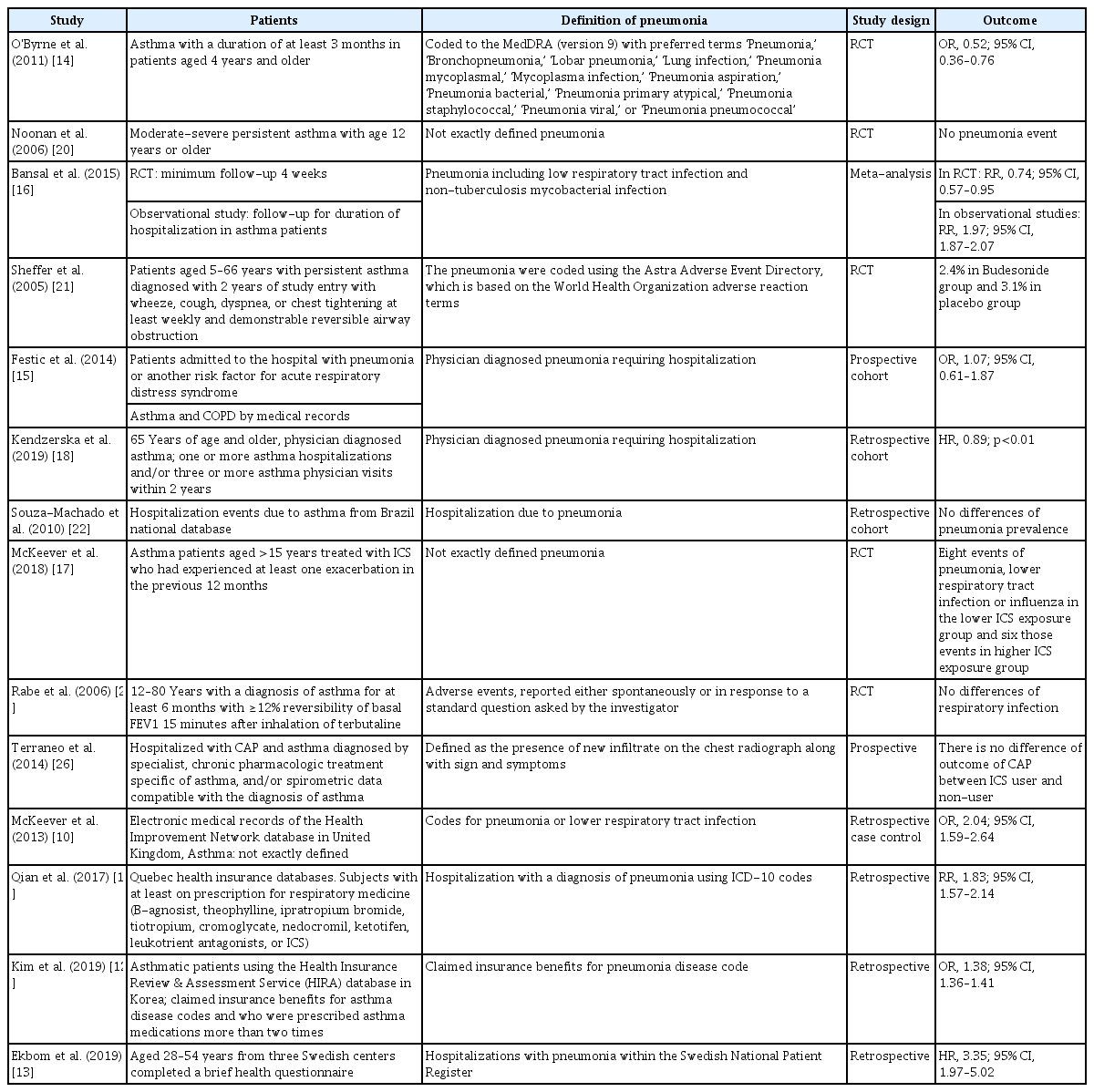
O’Byrne et al. [14] demonstrated that the use of ICS does not increase the occurrence of pneumonia; instead, it exerts a protective effect (pneumonia adverse event: 0.5% for budesonide and 1.2% for placebo, p<0.001), although this finding was confirmed by a retrospective analysis of a double-blind RCT, which was not designed to show the prevalence of pneumonia. Nevertheless, this study included a total of 14,993 patients (9,067 ICS users and 5,926 placebo users), which is large enough to demonstrate that ICS is not a risk factor for pneumonia in patients with asthma. Another RCT that enrolled 596 patients with asthma (348 vs. 125 vs. 123, budesonide-, placebo-, and formoterol-containing regimen, respectively) demonstrated that there were no events of pneumonia in the ICS-containing regimen group [20]. In addition, in an RCT of more than 7,000 patients, pneumonia was not associated with the use of ICS [21].
Two meta-analyses also consistently showed that ICS use was not associated with the risk of pneumonia [16,19]. In a meta-analysis published in 2015, RCTs and observational studies were analyzed separately. The estimated overall risk of pneumonia with ICS use in RCTs was within the protective range. In contrast, a summed OR of 1.97 (95% CI, 1.87 to 2.07) demonstrated that ICS use was a risk factor for pneumonia in patients with asthma in observational studies [16]. Htun et al. [19] suggested that only observational studies, albeit many, have previously reported an increased risk of pneumonia, and that observational studies that excluded chronic obstructive pulmonary disease (COPD) did not show a relationship between the use of ICS and the risk of pneumonia in patients with asthma. The currently available evidence suggests that ICS use is not a risk factor for pneumonia in patients with asthma [19].
A multicenter prospective study conducted in the USA showed that ICS users had a higher prevalence of pneumonia than did non-ICS users among patients with asthma; however, after adjusting for demographics, comorbidities, and medication, ICS use was not a risk factor for pneumonia (OR, 1.07; 95% CI, 0.61 to 1.87) [15]. A Canadian retrospective longitudinal cohort study including patients with asthma and those with COPD aged 66 years or older analyzed ICS effectiveness and safety. The primary outcome was a difference in admission due to obstructive lung disease or pneumonia according to ICS exposure. A total of 87,690 patients with asthma were enrolled, and ICS exposure was found to be associated with fewer hospitalizations for pneumonia (hazard ratio, 0.89; p<0.001). In this study, to reduce the prevalence of user and selection biases, the authors only included ICS users with a preceding 1-year ICS-free period [18].
A large-scale public health intervention project investigating patients with severe asthma was conducted in Brazil. All patients were encouraged to participate in this project and were educated to use ICS-containing inhalers. After implementing this project, 220,889 ICS-containing inhalers were dispensed, an increase from nearly 0. As expected, the admission rate for acute exacerbation of asthma was reduced by up to 74%, and hospitalization for pneumonia did not increase [22].
Risk of Pneumonia with Cumulative Doses of ICS and between Different ICS Types in Patients with Asthma
There are limited data showing dose dependency for the development of pneumonia in patients with asthma. The Fourfold Asthma Study compared asthma control status between the usual care group and the group that was asked to temporarily quadruple the ICS dose when deterioration was experienced. This multicenter RCT enrolled 1,922 participants and showed no difference in events for pneumonia, lower respiratory tract infection, or influenza between the two groups despite the use of higher cumulative doses of ICS in the intervention group [17]. Single maintenance and reliever therapy is recommended in steps 3 and 4 of asthma, and several recent studies have shown that it is effective in mild asthma [23,24]. Beasley et al. [24] demonstrated that the budesonide-formoterol (BF) used as-needed group showed superior outcomes to those of the albuterol used as-needed group and non-inferior outcomes to those of the budesonide maintenance group for preventing asthma exacerbations in adults with mild asthma. As expected, the cumulative ICS dose was higher in the budesonide maintenance group than in the BF group. However, there were no differences in any adverse events related to respiratory tract infections. One RCT further showed the efficacy and safety of fluticasone furoate (FF), compared with a placebo, in persistent asthma according to the FF dose. This study revealed that FF at total daily doses of 200 or 400 μg was significantly more effective than placebo for improving lung function, and the incidence of bronchitis, cold, influenza, or respiratory tract infection was comparable in both FF groups. In contrast, two retrospective studies showed that high-dose ICS was associated with an increased risk of pneumonia. One is an observational study using data from the health insurance database in Quebec; it showed that high-dose ICS was associated with a 96% increase in pneumonia risk, whereas lowdose ICS was associated with a 60% increase in risk. However, considering that the risk of pneumonia was lower in the medium-dose group (relative risk [RR], 1.53) than in the low-dose group (RR, 1.60), even this study failed to show a trend toward an increased risk of pneumonia according to the ICS dose [11]. The other study demonstrated that compared with fluticasone ≤250 µg, fluticasone >250 µg was associated with an increased risk of lower respiratory tract infection or pneumonia, whereas there was no clear dose-response relationship in other type of ICS [10].
Other studies have shown that the clinical course of pneumonia does not differ according to cumulative doses of ICS. In Japan, patients with asthma hospitalized for CAP between 1989 and 2001 have been studied. Researchers compared the patients’ body temperature, laboratory results, and resolution period according to the ICS dose (<800, 800, and >800 µg/day), finding no significant differences in these characteristics [25]. Another study evaluated clinical outcomes and the microbiology of CAP in patients with asthma. This prospective study showed that patients with asthma who used ICS more frequently had less severe pneumonia measured using the pneumonia severity index, CURB-65 (confusion, blood urea nitrogen, respiratory rate and blood pressure with age [65 years]), and partial oxygen pressure/fraction of inspired oxygen ratio and a shorter length of stay. The microbiological outcome, mortality, or use of mechanical ventilation in patients with asthma were similar to those of the general population without asthma [26]. Severe acute respiratory syndrome coronavirus 2 infection was also not associated with ICS in terms of prior use, doses, or types [27].
There has been no head-to-head comparison of budesonide and fluticasone in an RCT for the risk of pneumonia in patients with asthma. However, two observational studies have evaluated whether there is a difference in hospitalization rates for pneumonia according to the use of fluticasone propionate or budesonide [11,13]. One study performed in Canada showed that both fluticasone and budesonide were associated with an increased risk of pneumonia; the other study performed in Sweden demonstrated that the administration of fluticasone propionate was a risk factor for pneumonia, whereas no significant association was found with the administration of budesonide. However, Swedish study enrolled patients with asthma who completed only a brief questionnaire for self-reported diagnosis of asthma; thus, they could not exclude patients with COPD, and the risk of selection bias was high [13].
Limitations of Studies Suggesting an Association between ICS and the Increased Risk of Pneumonia in Asthma
As mentioned above, four studies have suggested that the use of ICS is associated with an increased risk of pneumonia in patients with asthma-like COPD [10-13]. All these studies were retrospective or cross-sectional and had a high risk of reverse causality; in the case of studies in which a large number of patients were analyzed, all used health insurance data [28]. In one study in the UK, cases of pneumonia hospitalization were not included. In addition, the primary outcome was defined as pneumonia or lower respiratory tract infection, and patients other than those with pneumonia were likely to be included [10]. Kim et al. [12] demonstrated that ICS use is a risk factor for pneumonia (OR, 1.38; 95% CI, 1.36 to 1.41); however, this study included long-acting muscarinic antagonist (LAMA) users, indicating a chance of selection bias as COPD was not excluded. Another Swedish cohort could not exclude the risk of COPD because self-reported diagnoses of asthma and pneumonia events were too low to have statistical power [13]. Qian et al. [11] included only individuals aged 12 to 35 years and LAMA users. In addition, they defined ICS users as those who used ICS within 60 days prior to pneumonia, but the reason for ICS use was not suggested in this manuscript; therefore, the risk of participant selection bias was high.
The studies in which there was no relationship between the use of ICS and pneumonia in asthma also had limitations. First, the primary outcome was not the prevalence of pneumonia according to ICS use [14,23,24]. Second, the definition of pneumonia varied. Third, selection bias was present during the randomization process of those studies whose sample size was too small. However, the number of enrolled patients in the RCTs showing the protective effect of ICS use against pneumonia limitations was sufficiently large. Table 1 summarizes the previous studies that evaluated the risk of pneumonia with ICS use as the primary outcome.
Conclusion
Several observational retrospective studies showing ICS use as a risk factor for pneumonia among patients with asthma had many limitations, including a high risk of selection bias and confounding factors. Therefore, these studies did not show causality.
In contrast, RCTs with a sufficient number of patients reported that the use of ICS reduced the risk of pneumonia, and other prospective design studies showed that ICS use in asthma did not increase the occurrence of pneumonia. Therefore, ICS is not associated with a risk of pneumonia in patients with asthma.
Notes
Authors’ Contributions
Conceptualization: Lee YJ. Methodology: Lee YJ. Investigation: Lee YJ. Writing - original draft preparation: Lee YJ. Writing - review and editing: Lee YJ, Park YB. Approval of final manuscript: all authors.
Conflicts of Interest
No potential conflict of interest relevant to this article was reported.
Funding
No funding to declare.