1. Bousquet J, Jeffery PK, Busse WW, Johnson M, Vignola AM. Asthma: from bronchoconstriction to airways inflammation and remodeling. Am J Respir Crit Care Med 2000;161:1720-45.


2. Chung KF, Godard P, Adelroth E, Ayres J, Barnes N, Barnes P, et al. Difficult/therapy-resistant asthma: the need for an integrated approach to define clinical phenotypes, evaluate risk factors, understand pathophysiology and find novel therapies. ERS Task Force on Difficult/Therapy-Resistant Asthma. European Respiratory Society. Eur Respir J 1999;13:1198-208.


3. Frey U. Predicting asthma control and exacerbations: chronic asthma as a complex dynamic model. Curr Opin Allergy Clin Immunol 2007;7:223-30.


4. DiMango E, Rogers L, Reibman J, Gerald LB, Brown M, Sugar EA, et al. Risk factors for asthma exacerbation and treatment failure in adults and adolescents with well-controlled asthma during continuation and step-down therapy. Ann Am Thorac Soc 2018;15:955-61.


6. Selroos O, Pietinalho A, Lofroos AB, Riska H. Effect of early vs late intervention with inhaled corticosteroids in asthma. Chest 1995;108:1228-34.


7. Drazen JM, Israel E, OŌĆÖByrne PM. Treatment of asthma with drugs modifying the leukotriene pathway. N Engl J Med 1999;340:197-206.


8. Kim DK, Park YB, Oh YM, Jung KS, Yoo JH, Yoo KH, et al. Korean Asthma Guideline 2014: summary of major updates to the Korean asthma guideline 2014. Tuberc Respir Dis 2016;79:111-20.

9. Lai CK, De Guia TS, Kim YY, Kuo SH, Mukhopadhyay A, Soriano JB, et al. Asthma control in the Asia-Pacific region: the asthma insights and reality in Asia-Pacific study. J Allergy Clin Immunol 2003;111:263-8.


10. Hedlin G, Bush A, Lodrup Carlsen K, Wennergren G, De Benedictis FM, Melen E, et al. Problematic severe asthma in children, not one problem but many: a GA2LEN initiative. Eur Respir J 2010;36:196-201.


11. Kim SR, Lee YC, Sung MJ, Bae HW. Current epidemiological data on asthma management in South Korea from qualitative assessment of asthma management by Health Insurance Review and Assessment Service (HIRA). Tuberc Respir Dis 2017;80:221-5.

13. van de Veen W, Wirz OF, Globinska A, Akdis M. Novel mechanisms in immune tolerance to allergens during natural allergen exposure and allergen-specific immunotherapy. Curr Opin Immunol 2017;48:74-81.


14. Makhinova T, Barner JC, Richards KM, Rascati KL. Asthma controller medication adherence, risk of exacerbation, and use of rescue agents among Texas medicaid patients with persistent asthma. J Manag Care Spec Pharm 2015;21:1124-32.


15. Chou CL, Perng DW, Lin TL, Lin AM, Chen TJ, Wu MS, et al. Analysis of prescription pattern and guideline adherence in the management of asthma among medical institutions and physician specialties in Taiwan between 2000 and 2010. Clin Ther 2015;37:2275-85.


16. Laforest L, Licaj I, Devouassoux G, Chatte G, Belhassen M, Van Ganse E, et al. Relative exposure to controller therapy and asthma exacerbations: a validation study in community pharmacies. Pharmacoepidemiol Drug Saf 2014;23:958-64.


17. Dean BB, Calimlim BM, Kindermann SL, Khandker RK, Tinkelman D. The impact of uncontrolled asthma on absenteeism and health-related quality of life. J Asthma 2009;46:861-6.


18. Gold LS, Thompson P, Salvi S, Faruqi RA, Sullivan SD. Level of asthma control and health care utilization in Asia-Pacific countries. Respir Med 2014;108:271-7.


19. Meng YY, Babey SH, Hastert TA, Lombardi C, Brown ER. Uncontrolled asthma means missed work and school, emergency department visits for many Californians. Policy Brief UCLA Cent Health Policy Res 2008;(PB2008-2):1-8.
20. Barnes PJ, Jonsson B, Klim JB. The costs of asthma. Eur Respir J 1996;9:636-42.


21. Jacob C, Bechtel B, Engel S, Kardos P, Linder R, Braun S, et al. Healthcare costs and resource utilization of asthma in Germany: a claims data analysis. Eur J Health Econ 2016;17:195-201.


22. Sadatsafavi M, Rousseau R, Chen W, Zhang W, Lynd L, FitzGerald JM. The preventable burden of productivity loss due to suboptimal asthma control: a population-based study. Chest 2014;145:787-93.


24. Williams SA, Wagner S, Kannan H, Bolge SC. The association between asthma control and health care utilization, work productivity loss and health-related quality of life. J Occup Environ Med 2009;51:780-5.




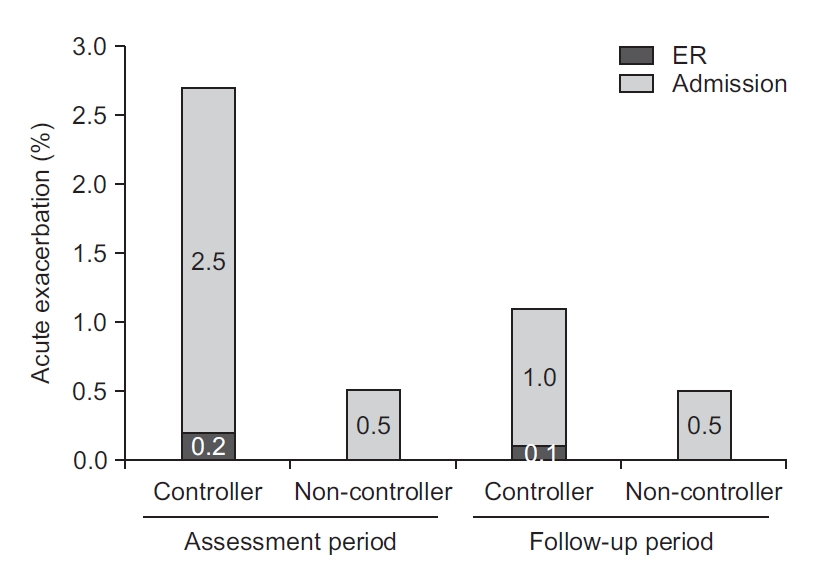
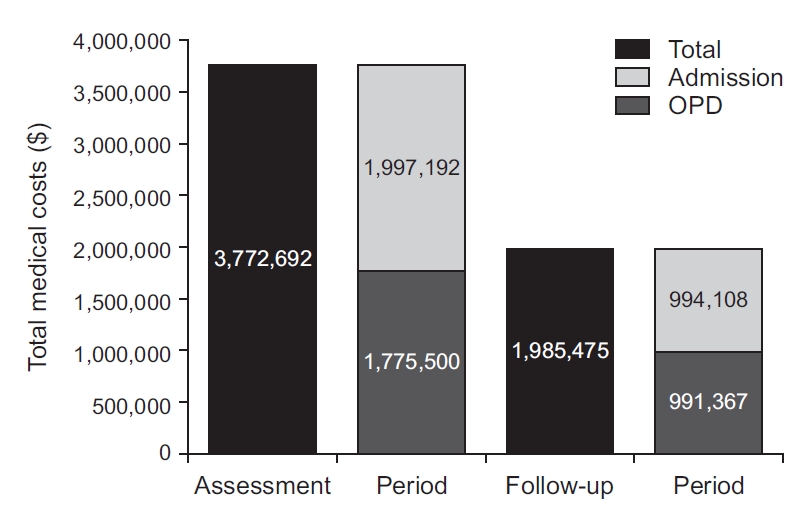
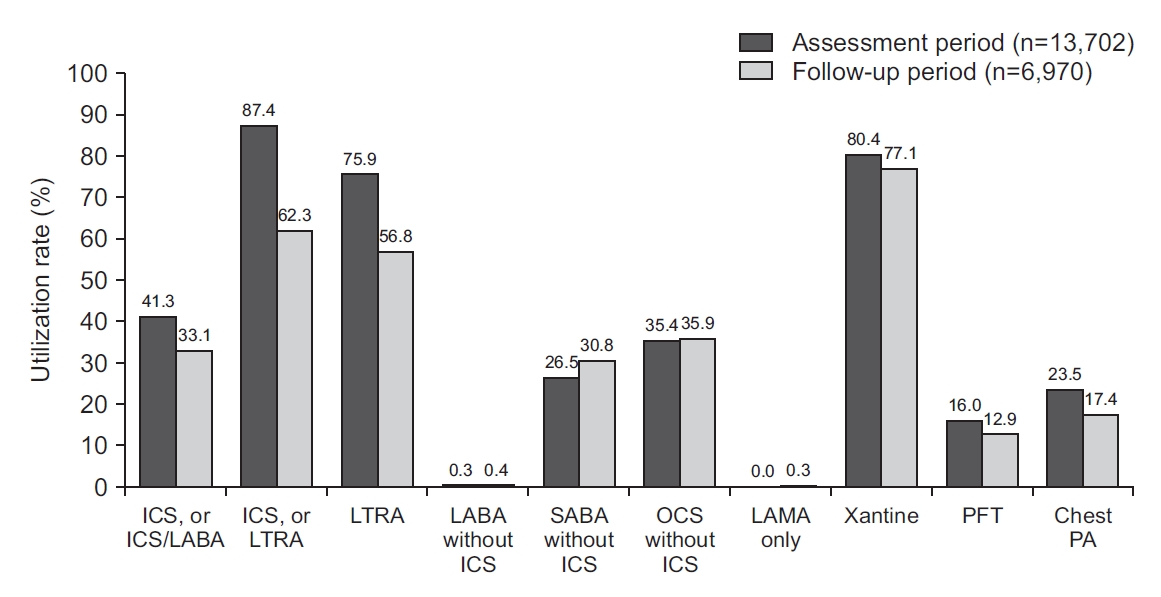





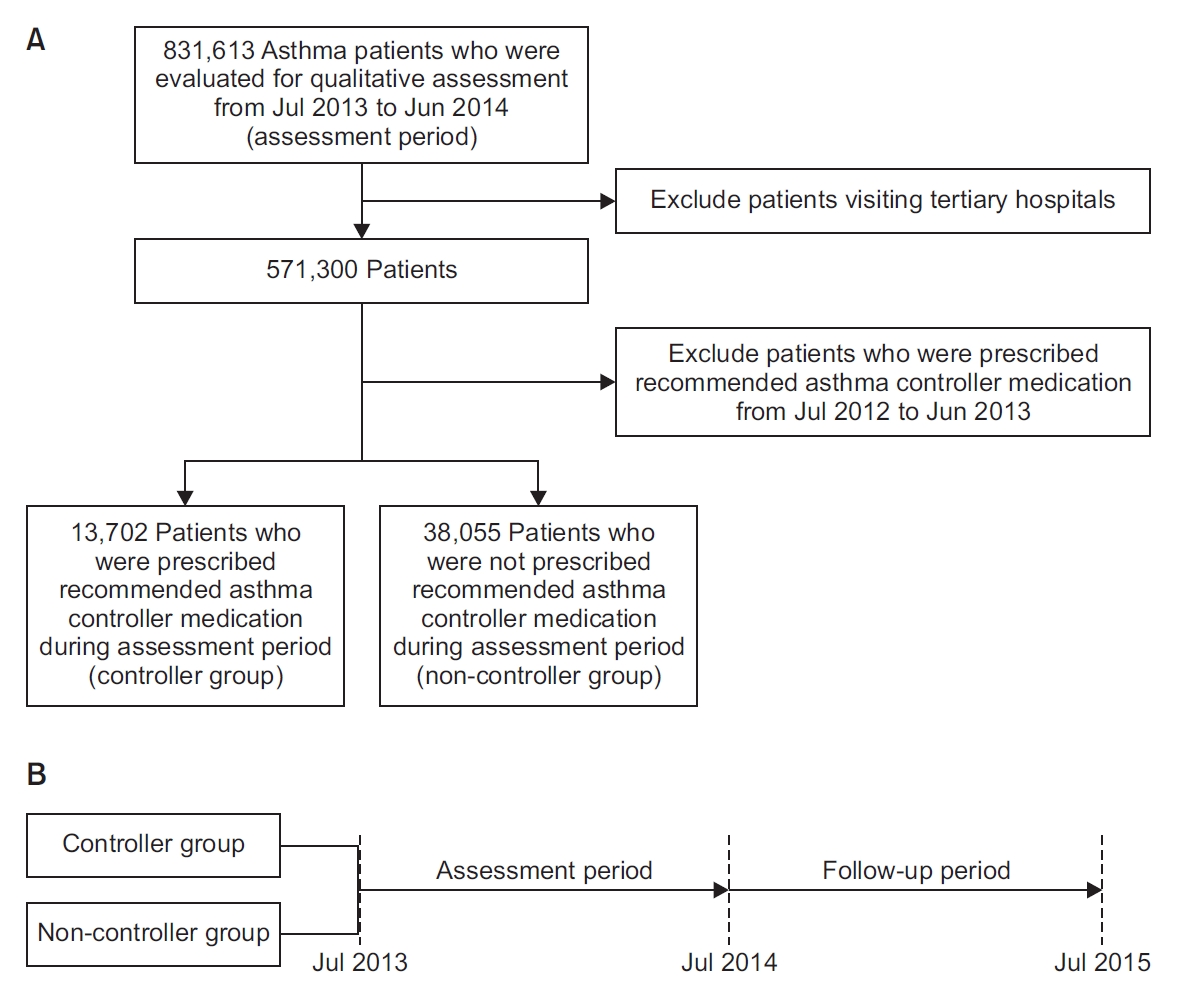
 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation



