 |
 |
| Tuberc Respir Dis > Volume 76(6); 2014 > Article |
|
Abstract
Background
We aimed to investigate the role of the physician in practice and the factors that influence the success rate of smoking cessation.
Methods
This study retrospectively analyzed 126 adult smokers who had visited the outpatient department of pulmonology, and received motivational interviewing with or without supplement drugs. The findings include continuous smoking abstinence rate, which was evaluated at 6, 12 and 24 weeks, and the factors associated with continuous abstinence for 6 months or longer.
Results
The patients with only motivational interviewing accounted for 57.9%, while the nicotine patch therapy was applied to 30.2%; and varenicline was prescribed to 11.9%. The smoking cessation success rates of at 6, 12, and 24 weeks were 55.6%, 47.6%, and 33.3%, respectively. However, even in the failure group at six months, tobacco consumption was decreased under 10 cigarettes per day in 42.1% (53/126). In multivariate logistic regression analysis, degree of Fagerstöm Test for Nicotine Dependence (p=0.034; odds ratio, 3.607; 95% confidence interval [CI], 1.102-1.807), the absence of smoking-related lung disease (p=0.008; odds ratio, 4.693; 95% CI, 1.497-14.707), and education level (p=0.001; odds ratio, 181.420; 95% CI, 8.414-3,911.502) were the predictors of successful smoking cessation.
Tobacco smoking is an important contributor to respiratory disease, including chronic obstructive pulmonary disease (COPD) and lung cancer, cardiovascular disease, and cancers of various organs. Tobacco smoking also negatively affects several other respiratory diseases, including pneumonia and pulmonary tuberculosis1. Therefore, smoking cessation is a practical way to prevent and treat smoking-related disease. In Korea, the adult male smoking rate was 66.3% in 1998; however, increased interest in health and a smoking cessation promotion policy, resulted in a reduction in this figure to 47.7% in 2008. However, that has been at a standstill since 2008, with 47.3% in 20112. The reason for the high smoking rate among males, despite the well-known harmfulness of smoking, is attributable to addiction caused by nicotine contained in cigarettes. Thus, it is necessary to perceive smoking as a disease that should be managed and treated as a drug addiction or chronic diseases rather than as a personal habit or hobby3.
Smoke cessation programs currently in effect are divided into pharmacotherapies and behavioral therapies or combinations thereof. Effective pharmacotherapies for smoking cessation include nicotine replacement therapy (NRT), bupropion or nortriptyline, and varenicline. Use of these forms of therapies increases quit rates 1.5- to 2.5-fold, and is a potentially valuable adjunct to any advice provided4. Until now, most domestic research on the effects of smoking cessation programs focused on effectiveness of the pharmacotherapy and targeted healthy adults who have decided to quit smoking5. Brief advice or counseling given by physicians to patients can strong and cost-effective smoking cessation program, can reduce morbidity and mortality4. Unfortunately, many smokers who see physicians do not receive assistance to quit. Accordingly, this study was conducted on patients who were encouraged to quit smoking, offered advice, and provided manners for smoking cessation by a pulmonologist at every outpatient session to investigate the 6-month continuous abstinence rate and to analyze the factors that influence the success rate.
This study was conducted on 126 subjects who visited the Division of Pulmonology of Seoul Medical Center from May 2011 to March 2012. All 126 subjects had been smoking more than five cigarettes per day and followed up for more than six months. The self-reported questionnaire with the Fagerstöm Test for Nicotine Dependence (FTND) was administered to the daily smokers at first visit. The FTND has six items with an overall score ranging between 0 and 10. In this study, high dependence was defined as a FTND score≥8 (low 0-3, moderate 4-7)6,7. The standardized questionnaire was included with their smoking history (including pack-years, starting age, previous quitting attempts, smoking-related and unrelated diseases, and motivational aspects of quitting), alcohol habit, educational level, marriage status, number of family members, pulmonology history (chief complaint at the time of the first visit, admission or not, final diagnosis and pulmonary disease status), and presence of underlying diseases including cardiovascular diseases, hypertension, diabetes, chronic kidney disease, malignancy and psychiatric diseases. Subject's height, weight, body mass index (BMI), pulmonary function test and chest radiograph were taken. Also, in the group aged >50 years and >20 pack-years were taken low dose chest computed tomography (CT) scans for lung cancer screening.
A pulmonologist provided motivation of smoking cessation according to modified forced expiratory volume at 1 second (FEV1) percentage by patient's age and disease status. The pulmonologist also presented a short lecture on nicotine dependence; syndromes of nicotine withdrawal; harmful effects of smoking and the benefits of quitting; how to handle weight gain and how to avoid trigger situations. The subjects were educated to know that smoking is a disease caused by nicotine addiction via multimedia and were provided with a medical handbook about smoking cessation made by our hospital. At each visit during the study period, motivational aspects, identification of triggers, management of weight gain, and use of medication were discussed for a mean time of 10 minutes. In addition, if the patient agrees, additional counseling for more than 30 minutes at first visit, telephone follow-up, and regular advice were also provided by the trained practical nurse. Regardless of this, all participants received one of three interventions; 1) motivational interviewing (MI) only, 2) MI with NRT-free offer, or 3) MI with varenicline (Figure 1).
The primary outcome considered was continuous smoking abstinence rate, which was evaluated at 6, 12, and 24 weeks. Smoking cessation success was defined as a subject orally reporting that he or she had not smoked at all for 6 months or longer, and for whom the result of exhaled CO level was consistently <10 ppm. We also studied the factors associated with continuous abstinence for 6 months or longer, including age, sex, educational level, marriage status, nicotine dependence, number of cigarettes smoked per day, and comorbidities. This study was approved by the Institutional Review Board at Seoul Medical Center. All subjects provided written informed consent.
Data analyses were performed using SPSS version 18.0 (SPSS Inc., Chicago, IL, USA). Pearson's chi-square test for categorical variables and a two sample t-test for continuous variables were used to test the baseline differences between the success and failure groups. A logistic regression model was used for the analysis of factors affecting the success of 6-month smoking cessation. The level of significance was established at α=0.05.
A total of 126 subjects entered the study from May 2011 to April 2012. The baseline characteristics of the study population are displayed in Table 1. The mean age of the subjects was shown to be 55.9 years (range, 19-78 years), comprising 111 (88.1%) males and 15 (11.9%) females. Eighty-three subjects with medical insurance (65.9%), 38 subjects with medical assistance (30.1%), and five subjects were homeless (4.0%).
The chief complaint at the time of the first visit was mainly dyspnea in 46 (36.5%), abnormal findings in regular examination in 23 (18.3%), and chronic cough in 19 (15.1%); the rest were diagnosed with pneumonia or tuberculosis in 12 (9.5%), increased sputum in 12 (9.5%), chest pain in 6 (4.8%), hemoptysis in 5 (4.0%), and upper respiratory infection in 3 (2.4%). Seventy-nine patients (62.7%) were diagnosed with smoking-related pulmonary disease (COPD, n=37, 29.4%; bronchial asthma, n=27, 21.4%; asthma COPD overlap syndrome [ACOS], n=9, 7.1%; chronic bronchitis, n=6, 4.8%) (Figure 2), and two each of the patients was diagnosed with interstitial lung disease and solitary pulmonary nodule. The rest of the patients were not diagnosed with lower respiratory tract disease. One hundred-eleven of 126 patients underwent pulmonary function test, the mean FEV1 was 64.9±20.1% (range, 17.0-110.0%) and the mean FEV1/forced vital capacity was 70.5±11.9% (range, 33.0-94.0%). Apart from pulmonary disease, six patients had cardiovascular disease (4.8%), 36 hypertension (28.6%), 25 diabetes mellitus (19.8%), and 15 psychiatric disorder, such as depression, anxiety disorder, and schizophrenia, were included in the study. The level of education was not high in these patients, 13 (10.3%) equal to or higher than college graduate, a high school diploma in 47 (37.3%), a middle school diploma in 55 (43.7%), and elementary school education or less in 11 (8.7%). Their total smoking time was 33.4±13.6 years (range, 5-60 years), mean tobacco consumption was 21.4±8.3 cigarettes per day (range, 4-40 cigarettes per day). Nicotine dependency was measured with the FTND questionnaire, and the mean score was 5.2±2.8.
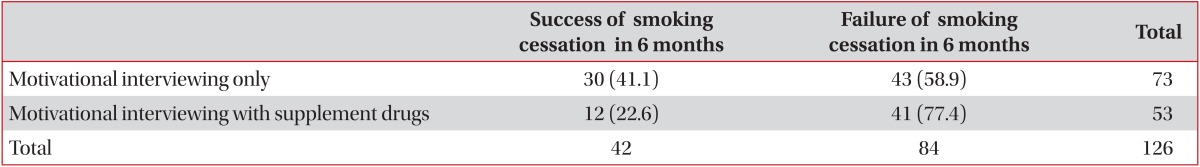
All the subjects performed motivational interviewing via pulmonologist during every consultation session, and 73 subjects (57.9%) had only motivational interviewing without pharmacotherapy. NRT was applied free of charge to 38 subjects (30.2%), and varenicline was prescribed to 15 subjects (11.9%); however, only seven subjects took varenicline for longer than 2 weeks (median, 3 weeks; range, 1-12 weeks). Thirty-two subjects agreed to have counseling with a practice nurse for smoking cessation; the average number of counseling was 2.7±0.44 (range, 1-11 times).
The success rate of 6-week smoking cessation of the 126 patients was shown to be 55.6% (70 patients), 47.6% (60 patients) for 3-month smoking cessation, was reduced to be 33.3% (42 patients) for 6-month smoking cessation (Table 1). However, even in the continuous smoking group, tobacco consumption was decreased to under 10 cigarettes per day in 53 patients (42.1%) among 126 patients, and in 28 patients (22.2%), tobacco consumption was kept under 5 cigarettes per day. Their average tobacco consumption was reduced from 21.4±8.3 cigarettes per day at the time of the first visit to 8.2±9.1 cigarettes per day after 6 months. According to the paired-samples t-test, the decrement was an average 13.2±9.7 cigarettes per day (p=0.000). The majority of smokers after continuous cessation of 6 weeks had relapsed at average 2.6±1.1 months; and smokers after continuous cessation of 3 months at an average of 3.3±0.6 months. In the 6-month smoking cessation success group, the mean FTND was 4.5±3.0, which was lower than 5.5±2.6 in the failure group; however, statistically significant differences between the two groups was not found (p=0.061). Nevertheless, according to the FTND severity, 45.2% of the group of 6-month smoking cessation was mild, 31.0% moderate, and 23.8% severe, and the differences were considered significant (p=0.013).
Thirty-six (38.3%) of the 94 subjects who had a will to quit smoking at first visit quit smoking; by comparison, only six (18.8%) of the 32 subjects who did not have a will to quit smoking. The number of family living together (p=0.510) and presence of spouse (p=0.693) did not show statistically significant differences. Twenty-eight subjects (66.7%) of the 42 subjects with 6-month smoking cessation were married, whereas 14.3% (6/42) were divorced. Only two of the 24 patients who were unmarried quit smoking, on the contrary, six of the seven who were bereaved did so. History of alcohol ingestion showed statistically significant difference in the group of 3-month smoking cessation (p=0.033), but not in the group of 6-month smoking cessation (p=0.428). The level of education showed statistically significant differences; only 16 of 66 (24.2%) who had less middle school diploma quit smoking, in comparison to 26 of 60 (43.3%) who had high school-level or higher education (p=0.019). BMI of group of 6-month smoking cessation (22.1±4.6) was lower than that of failure group (at six months) (23.3±3.5), through not statistically significant (p=0.112).
Chief complaint or diagnosis at the time of the first visit did not show statistically significant differences between two groups (p=0.439). In the group diagnosed with smoking-related lung disease such as COPD, bronchial asthma, and ACOS, the smoking cessation rate was 27.2% (22/81), which was significantly lower than that of 44.4% (20/45) in the group without smoking-related lung disease (p=0.039). Smoking cessation rate in patients with cardiovascular disease, hypertension, and diabetes did not show statistically significant differences. Only one (6.7%) of the 15 patients who had psychiatric disorder such as depression, anxiety disorder, and schizophrenia succeeded in quitting smoking, compared with 41 (36.9%) of the 111 patients who did not have psychiatric disorders (p=0.014).
There was no difference of smoking cessation rate according to the type of insurance (p=0.471), with 27 of 83 with medical insurance (32.5%), 2 of 5 homeless (40.0%), and 13 of 38 with medical assistance (34.2%). By paired-samples t-test, the mean FEV1 has significantly improved over the 3 months for both smoking cessation success group (10.9±15.1%) and the failure (at 3 months) group (6.8±11.2%); thus, it is likely due to treatment with medication of smoking-related lung disease. The patients with history of hospitalization showed more favorable results in the smoking cessation (p=0.003); 25 (48.1%) of the 52 patients who had history of admission during the follow-up period.
In the group of severe degree of FTND, prescription of varenicline was preferentially recommended; however, only 19 of the 126 patients accepted the prescription of varenicline, and only 15 patients actually took it for more than one day, because of the drug cost, side effects, and other reasons. The smoking cessation rate (33.3%, 5/15, p=0.623) of the 15 patients prescribed varenicline was disappointing; however, in the seven patients who took the medication for more than 2 weeks, the success rate was higher (71.4%, 5/7, p=0.041). In the motivational interviewing only group, smoking cessation rate was 41.1%, significantly higher than the NRT or varenicline group (22.6%; p=0.030) (Table 2).
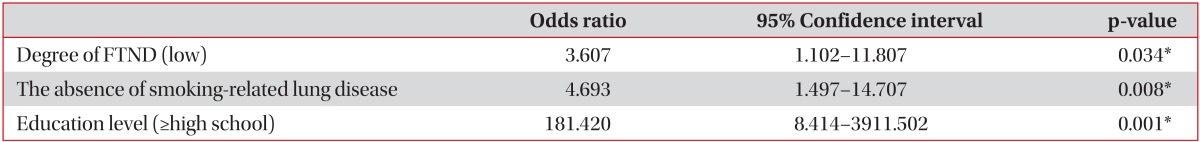
As a result of univariate analysis, factors affecting the success rate of 6-month smoking cessation were shown to be degree of FTND, mean number of cigarette smoking, a will to quit smoking, marriage status, the history of admission, the absence of smoking-related lung disease, history of psychiatric disease, higher educational level, and the prescription of supplement drug (varenicline>2 weeks). A multivariate analysis showed that the odds ratio of 6-month smoking cessation success increased to approximately 3.607-fold, 4.693-fold, and 181.420-fold in the cases of low degree of FTND, the absence of smoking-related lung disease, and higher educational level, respectively, if other factors were constant (Table 3).
Tobacco smoking is an important contributor to respiratory diseases; it is the major etiological factor for the development of COPD and lung cancer, and adversely affects control of asthma1. Smoking cessation is the most important intervention in COPD and respiratory disease, so it is important that pulmonary physicians act to ensure that patients with COPD and other respiratory diseases quit smoking.
In present study, the continuous smoking abstinence rates was 55.6%, 47.6%, and 33.3% at 6 weeks, 3 months, and 6 months, respectively. In one randomized clinical trial of transdermal nicotine patches in Korea, contrary to the point prevalence abstinence rates (20.3% at 3-month and 24.6% at 6-month follow-up), the continuous abstinence rates decreased linearly as follows: 20.3% at 3-month follow-up and 17.8% at 6-month follow-up8. The other studies with varenicline in Asia revealed that their success rates of 6-month smoking cessation were 37.7%9, 46.8%10, and 50.3%11. However, these randomized, placebo-controlled trials may not reflect the clinical practice, and in smokers that had actually visited an outpatient department, the motivation and readiness to quit could be different from smokers at smoking cessation clinics. Other researchers have estimated that only 3-5% of smokers are able to achieve prolonged abstinence for 6-12 months after a given unassisted quit attempt, an estimate consistent with the high levels of failure12.
The subjects of this study were patients seeking an outpatient department of pulmonology, with relatively low education level (equal to and lower than those of a middle school diploma in 52.4%), and low income level (34.1% were patients with medical assistance and homeless) from a socioeconomic point of view, and with brief smoking cessation intervention program in practice with low rate of prescribing medications (only 11.9% of patients used varenicline). Thus, it is likely that the success rate of 6-month continuous smoking abstinence was not low in the subjects of this study due to more experiences in abnormal symptoms and signs, and stronger motivation for smoking cessation by diagnosing of disease or having biofeedback of their FEV1 %.
One promising approach to encourage cessation among less motivated smokers is motivational interviewing13. Motivational interviewing is a patient-centered directive approach to enhance intrinsic motivation to behavioral change by helping patients explore and resolve ambivalence between the desired behavior and their actual behavior14,15. It focuses on what patients can do to improve their own health, as opposed to health care providers telling them what to do14. Ojedokun et al.16 reported that, in addition to brief cessation support during routine consultations, providing lung age biofeedback to smokers along with pharmacotherapy significantly increased (control 12.0% vs. intervention 22.1%), the proportion who quit within a month. In the present study, when promoting smoking cessation during clinical consultations, we provided the 'percentage of FEV1 compared with predictive value based on their age, height and ethnicity' biofeedback to patients, and 'extent of emphysema and bronchial wall thickening' also to the patients who underwent low-dose CT scans of chest.
However, in the present study, the degree of FEV1 % or the presence of abnormal findings on chest low-dose CT scans were not significant factors affecting the success rate of 6-month smoking cessation. We suggest that these factors could be helpful in motivational aspects of smoking cessation, but not in maintenance of cessation of smoking. Nevertheless, it is widely believed that planning for a difficult task like quitting smoking should result in increased success17. In addition, there is some evidence that smokers may be more receptive to advice to stop when it is linked with an existing medical condition (not necessarily smoking-related)18. However, until now, encouraging and supporting smoking cessation appears to be a low priority among physicians, even though this intervention is highly cost-effective and is the only intervention that reduces the risk of developing respiratory diseases and slows its progression. In this study, we were able to achieve the favorable continuous smoking abstinence rate by motivational interviewing with screening or diagnosing tool for lung diseases and continuous brief support for smoking cessation during every routine consultations. In some healthcare settings, they can also refer people to more intensive behavioral counseling and support, either face-to-face or via telephone quit line services4,19. However, the present study did not convincingly demonstrate that more intensive counseling from a practice nurse could increase cessation rates amongst smokers.
Several studies have shown that the main predictors of smoking cessation are age, gender, the daily consumption of tobacco, marital status (living with a spouse or partner), stress, social status, baseline motivation to stop smoking, nicotine dependency (FTND), and so on20,21. In the univariable logistic regression analysis of our study, we found that the number of cigarettes per day, degree of FTND, a will to quit smoking, the absence of psychiatric disease and smoking-related lung disease, the history of admission, education level, and prescription of varenicline and NRT were significant predictors for smoking cessation at 6-month follow-up. In this study, we could not check the income level of the patients; however, there was no difference according to the type of insurance or homeless status. Nevertheless, the patients with history of hospitalization showed more favorable results of the smoking cessation, because hospitalization may be a potentially powerful "teachable moment" in which smokers are often motivated to quit and are receptive to assistance due to concerns about their health22.
Although the prescription of varenicline or NRT was a factor related to 6-month smoking cessation, no significant difference was found in multivariate logistic regression analysis. Data from numerous randomized controlled trials demonstrate clearly the effectiveness of NRT, bupropion and varenicline in promoting long-term abstinence from smoking23. Possible explanation for the finding of lower effectiveness in the "real practice" is that many smokers fail to adhere to treatment recommendations23,24. Particularly, NRT was given free of charge every 2 weeks; it might be helpful for motivational aspect, through it was possible that the patients spontaneously discontinued treatment. In addition, for varenicline, due to drug cost, adverse events, and drug incompliance, 12-week varenicline treatment was rarely conducted, especially in public hospital with patients with low economic status. In fact, only 7 subjects (of 15) took varenicline for longer than 2 weeks (median, 3 weeks; range, 1-12 weeks) through their success rate was higher (5 of 7, 71.4%) than the 0% of the group that terminated varenicline treatment early.
In multivariate logistic regression analysis, degree of FTND, the absence of smoking-related lung disease, and education level were the predictors of successful smoking cessation. The FTND, a non-invasive and easy-to-obtain self-reported tool that conceptualizes dependence through physiological and behavioral symptoms, is used for assessing nicotine dependence7. In our study, the majority of the patients in the 6-month smoking cessation group had low to moderate (0-7) dependency in the FTND. In addition, smoking cessation rate was much lower in the patients with smoking-related lung disease. Although patients with respiratory disease have a greater and more urgent need to stop smoking than the average smoker, many often find it more difficult to do so. In one randomized controlled trial (RCT), 26 week-continuous abstinence rates of smokers with mild or moderate COPD were 16% in bupropion group, and 9% in placebo group25. Some smokers succeed in quitting and, thus, drop out of the smoking population, but the majority fail. It follows from this selection hypothesis that the more severe the lung disease, the more difficult it is to give up smoking1. Another factor that can contribute to the difficulties faced by respiratory patients who smoke is the relatively high prevalence of depression or low mood1.
Many smokers would prefer to reduce the number of cigarettes smoked daily rather than quitting completely. In several RCTs of smoking reduction, a reduction in daily cigarette smoking of 50% after 3-4 months had a strong predictive value for quitting at 1 year (a reduction in daily cigarette smoking in 15.9% of smokers using NRT vs. 6.7% using placebo → smoking cessation rates after 1 year in 8.4% among NRT users vs. 4.1% in placebo users)1. In this study, tobacco consumption was decreased to under 10 cigarettes per day in 42.1% (53/126), and in 22.2%, tobacco consumption was kept under five cigarettes per day. Their tobacco consumption decrement was an average of 13.2±9.7 cigarettes per day (21.4±8.3 at the time of the first visit → 8.2±9.1 after 6 months cigarettes per day). The smoking reduction concept should be offered to patients with respiratory disorders who smoke and who are not motivated to quit1. Smoking reduction seems to have a role for smokers not currently motivated or able to quit as a gateway to complete cessation; however, we cannot conclude that a reduction in daily cigarette smoking leads to smoking cessation at long term follow-up.
Additional limitation of this study include that it used data from only one outpatient clinic in public hospital; the results, therefore, need to be confirmed by more studies. In addition, this is a retrospective observational study that was conducted on patients seeking an outpatient department of pulmonology, there are many volatile variables, such as the number of visits, the timing of diagnosed of respiratory disease, and the use of respiratory medicines, etc. In addition, it does not analyze withdrawal symptoms or gained weight, and adverse events of prescribed drugs, and used dosage of NRT or varenicline in "real-world." As varenicline was not provided free of charge, subjects had to purchase them themselves on prescription, while NRT was provided free of charge. Additionally, the number of cigarettes of patients smoking per day was self-reported by questionnaire during every consultation session, and it remains possible that some exaggerated reporting occurred.
The findings reported here show that, in clinical practices, not in smoking cessation clinic, a disease management approach with motivational interviewing, associated or not with pharmacotherapy, was effective in helping smokers to quit. Even smokers who are initially unwilling to quit might be engaged in smoking cessation with a significant reduction in daily cigarette smoking. Physicians play a critical role in reducing the burden of tobacco-related health problems by helping their patients who smoke to quit and by motivating their nonsmoking patients to remain nonsmokers. Unfortunately, many physicians report inadequate training in smoking cessation, and many smokers who see physicians do not receive assistance to quit4. Advances can be made by not only improving the use of existing smoking cessation treatments but also intervention by physicians that increases motivation, self-efficacy and self-esteem.
References
1. Tonnesen P, Carrozzi L, Fagerstrom KO, Gratziou C, Jimenez-Ruiz C, Nardini S, et al. Smoking cessation in patients with respiratory diseases: a high priority, integral component of therapy. Eur Respir J 2007;29:390-417. PMID: 17264326.


2. Ministry of Health and Welfare. Korea National Health and Nutrition Examination Survey (KNHANES V-3). Seoul: Korea Centers for Disease Control and Prevention; 2012.
3. Lee JY, Kim MJ, Jun HJ, Kang M, Park AR, Oh DE, et al. Adherence to varenicline and abstinence rates for quitting smoking in a private health promotion center-based smoking cessation clinic. Tuberc Respir Dis 2012;72:426-432.

4. Stead LF, Buitrago D, Preciado N, Sanchez G, Hartmann-Boyce J, Lancaster T. Physician advice for smoking cessation. Cochrane Database Syst Rev 2013;5:CD000165PMID: 23728631.

5. Jeon K, Song JU, Um SW, Koh WJ, Suh GY, Chung MP, et al. Bronchoscopic findings of pulmonary paragonimiasis. Tuberc Respir Dis 2009;67:512-516.

6. Heatherton TF, Kozlowski LT, Frecker RC, Fagerstrom KO. The Fagerstrom test for nicotine dependence: a revision of the Fagerstrom Tolerance Questionnaire. Br J Addict 1991;86:1119-1127. PMID: 1932883.


7. Perez-Rios M, Santiago-Perez MI, Alonso B, Malvar A, Hervada X, de Leon J. Fagerstrom test for nicotine dependence vs heavy smoking index in a general population survey. BMC Public Health 2009;9:493PMID: 20042106.




8. Myung SK, Seo HG, Park S, Kim Y, Kim DJ, Lee DH, et al. Sociodemographic and smoking behavioral predictors associated with smoking cessation according to follow-up periods: a randomized, double-blind, placebo-controlled trial of transdermal nicotine patches. J Korean Med Sci 2007;22:1065-1070. PMID: 18162724.



9. Nakamura M, Oshima A, Fujimoto Y, Maruyama N, Ishibashi T, Reeves KR. Efficacy and tolerability of varenicline, an alpha4beta2 nicotinic acetylcholine receptor partial agonist, in a 12-week, randomized, placebo-controlled, dose-response study with 40-week follow-up for smoking cessation in Japanese smokers. Clin Ther 2007;29:1040-1056. PMID: 17692720.


10. Tsai ST, Cho HJ, Cheng HS, Kim CH, Hsueh KC, Billing CB Jr, et al. A randomized, placebo-controlled trial of varenicline, a selective alpha4beta2 nicotinic acetylcholine receptor partial agonist, as a new therapy for smoking cessation in Asian smokers. Clin Ther 2007;29:1027-1039. PMID: 17692719.


11. Wang C, Xiao D, Chan KP, Pothirat C, Garza D, Davies S. Varenicline for smoking cessation: a placebo-controlled, randomized study. Respirology 2009;14:384-392. PMID: 19192221.


12. Hughes JR, Keely J, Naud S. Shape of the relapse curve and long-term abstinence among untreated smokers. Addiction 2004;99:29-38. PMID: 14678060.


13. Catley D, Harris KJ, Goggin K, Richter K, Williams K, Patten C, et al. Motivational Interviewing for encouraging quit attempts among unmotivated smokers: study protocol of a randomized, controlled, efficacy trial. BMC Public Health 2012;12:456PMID: 22713093.




14. Noordman J. Lifestyle counseling by physicians and practice nurses in primary care: an analysis of daily practice. Utrecht: Netherlands Institute for Health Services Research; 2013.
15. Martins RK, McNeil DW. Review of motivational interviewing in promoting health behaviors. Clin Psychol Rev 2009;29:283-293. PMID: 19328605.


16. Ojedokun J, Keane S, O'Connor K. Lung age bio-feedback using a portable lung age meter with brief advice during routine consultations promote smoking cessation? Know2quit multicenter randomized control trial. J Gen Pract 2013;1:123.

17. Borland R, Balmford J, Swift E. Effects of timing of initiation and planning on smoking cessation outcomes: study protocol for a randomised controlled trial. BMC Public Health 2013;13:235PMID: 23496992.




18. West R, McNeill A, Raw M. Smoking cessation guidelines for health professionals: an update. Health Education Authority. Thorax 2000;55:987-999. PMID: 11083883.



19. Lancaster T, Stead L. Extended-duration transdermal nicotine therapy was more effective than standard-duration therapy for smoking cessation. Ann Intern Med 2010;152:JC4-JC8. PMID: 20404377.

20. Gourlay SG, Forbes A, Marriner T, Pethica D, McNeil JJ. Prospective study of factors predicting outcome of transdermal nicotine treatment in smoking cessation. BMJ 1994;309:842-846. PMID: 7950614.



21. Yeo CD, Kang HH, Kang JY, Kim SK, Kim MS, Kim SS, et al. A short-term effectiveness of smoking cessation intervention in outpatient department of pulmonology. Tuberc Respir Dis 2011;71:114-119.

22. Balmford J, Leifert JA, Schulz C, Elze M, Jaehne A. Implementation and effectiveness of a hospital smoking cessation service in Germany. Patient Educ Couns 2014;94:103-109. PMID: 24176610.


23. Raupach T, Brown J, Herbec A, Brose L, West R. A systematic review of studies assessing the association between adherence to smoking cessation medication and treatment success. Addiction 2014;109:35-43. PMID: 23919621.


24. Shah SD, Wilken LA, Winkler SR, Lin SJ. Systematic review and meta-analysis of combination therapy for smoking cessation. J Am Pharm Assoc (2003) 2008;48:659-665. PMID: 18826906.


25. Tashkin D, Kanner R, Bailey W, Buist S, Anderson P, Nides M, et al. Smoking cessation in patients with chronic obstructive pulmonary disease: a double-blind, placebo-controlled, randomised trial. Lancet 2001;357:1571-1575. PMID: 11377644.


Figure 1
Intervention for smoking cessation of participants through the study. MI: motivational interviewing; NRT: nicotine patch therapy.

Figure 2
The main diagnosis of respiratory disease on enrollment. COPD: chronic obstructive pulmonary disease; TB: tuberculosis; ACOS: asthma COPD overlap syndrome; SPN: solitary pulmonary nodule; ILD: interstitial lung diseas.




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation




