 |
 |
| Tuberc Respir Dis > Volume 78(4); 2015 > Article |
|
Abstract
Pulmonary arteriovenous malformations (AVMs) are caused by abnormal vascular communications between the pulmonary arteries and pulmonary veins, which lead to the blood bypassing the normal pulmonary capillary beds. Pulmonary AVMs result in right-to-left shunts, resulting in hypoxemia, cyanosis, and dyspnea. Clinical signs and symptoms vary depending on the size, number, and flow of the AVMs. Transcatheter embolization is the treatment of choice for pulmonary AVMs. However, this method can fail if the AVM is large or has multiple complex feeding arteries. Surgical resection is necessary in those kind of cases. Here, we report the case of a patient with a 6-cm pulmonary AVM with multiple feeding arteries that was successfully treated by repeated coil embolization without surgery.
Pulmonary arteriovenous malformations (AVMs) are caused by abnormal vascular communications between the pulmonary arteries and pulmonary veins, which lead to blood bypassing the normal pulmonary capillary beds. AVMs result in right-to-left shunts that subsequently cause hypoxemia1. Pulmonary AVMs vary in size from 1 to 5 cm. Generally, pulmonary AVMs <2 cm in size do not produce clinical symptoms2,3. Despite the lack of clinical symptoms in most cases, pulmonary AVMs usually require treatment due to the considerably high rates of morbidity and mortality associated with the condition.
Before the 1980s, surgical resection was the only available treatment for AVMs. Since then, advances in treatments have led to the successful application of transcatheter embolization. Currently, transcatheter embolization is the treatment of choice for pulmonary AVMs1. However, some pulmonary AVMs are not amenable to transcatheter embolization if they have a large size or are associated with multiple complex feeding arteries. Although surgical resection is occasionally recommended in patients with large pulmonary AVMs, no definite threshold for the surgical indication is present4,5. In the present report, we describe the case of a patient with a large (6 cm├Ś3 cm) pulmonary AVM that was successfully treated by repeated transcatheter embolization without surgery.
A 69-year-old man with a history of hypertension and pulmonary tuberculosis presented with chronic rhinorrhea and nasal obstruction. He was a former smoker (50 pack-year). He was diagnosed with chronic sinusitis and scheduled to undergo surgical treatment for sinusitis. He had undergone chest radiography during the preoperative evaluation. The radiography indicated a mass-like opacity in the right lower lung field (Figure 1), and the patient was referred to a pulmonologist. He denied any history of dyspnea or chest pain. There were no other respiratory symptoms such as cough, sputum production, or hemoptysis. On physical examination, his breathing sounds were normal and there were no abnormal lung sounds such as crackles, wheezing, or bruit. However, his lips and fingertips showed cyanotic changes, and there was clubbing in both fingers.
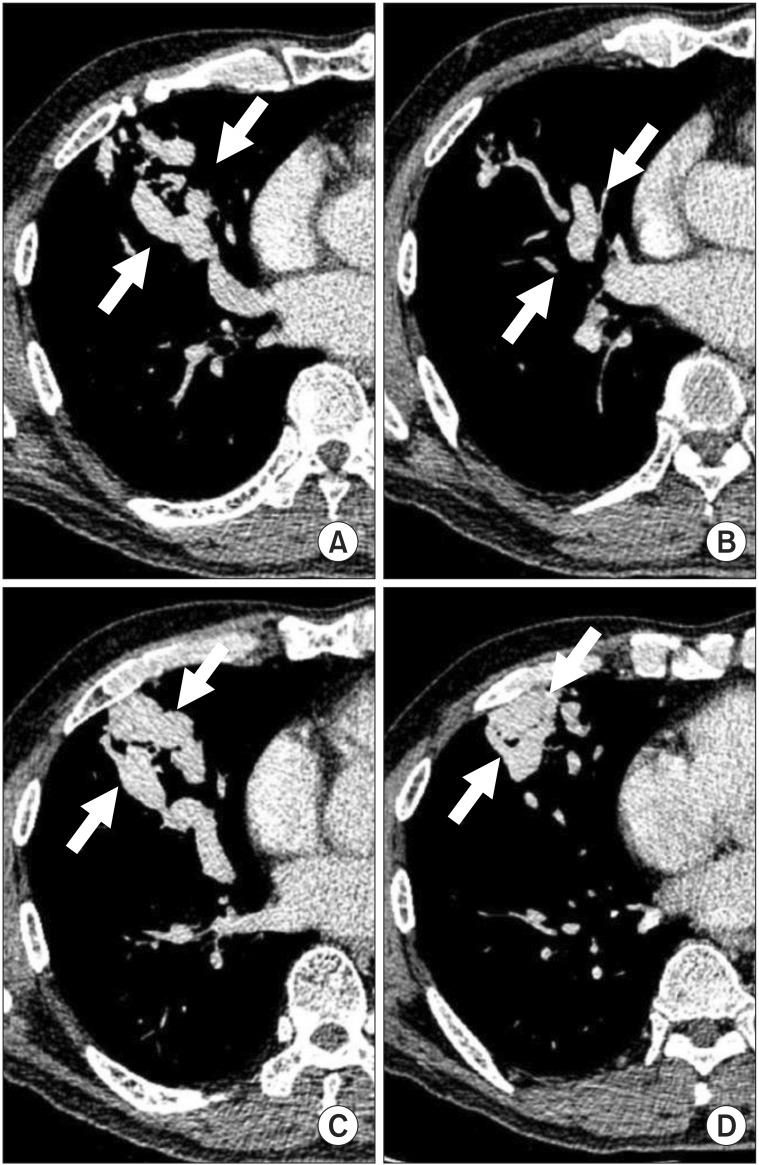
On admission, vital signs were within normal ranges, although hypoxia was detected by pulse oximetry. Initially, his SpO2 level on room air was 80%, and arterial blood gas analysis demonstrated hypoxemia with a PaO2 level of 52.1 mm Hg using a nasal prong at 3 L/min. Laboratory studies showed marked erythrocytosis. His white blood cell count was 5.31├Ś103/┬ĄL, hemoglobin level was 19.6 g/dL, and platelet count was 177├Ś103/┬ĄL. Contrast-enhanced chest computed tomography (CT) revealed a large (approximately 6 cm├Ś3 cm) entangled vascular lesion in the right middle lobe, and findings consistent with pulmonary AVM (Figure 2). The largest feeding vessel had a maximum diameter of 12 mm. The electrocardiography showed normal sinus rhythm without significant ST segment change.
The patient's skin, oral cavity, and nasal cavity were carefully examined, but there were not identified telangiectatic lesions. He had chronic rhinorrhea, but denied history of recurrent or spontaneous epistaxis. There were no family members who had been diagnosed with visceral AVMs before. As there were no evidences suggesting the diagnosis of hereditary hemorrhagic telangiectasia (HHT), this patient was regarded as having non-HHT-related pulmonary AVM.
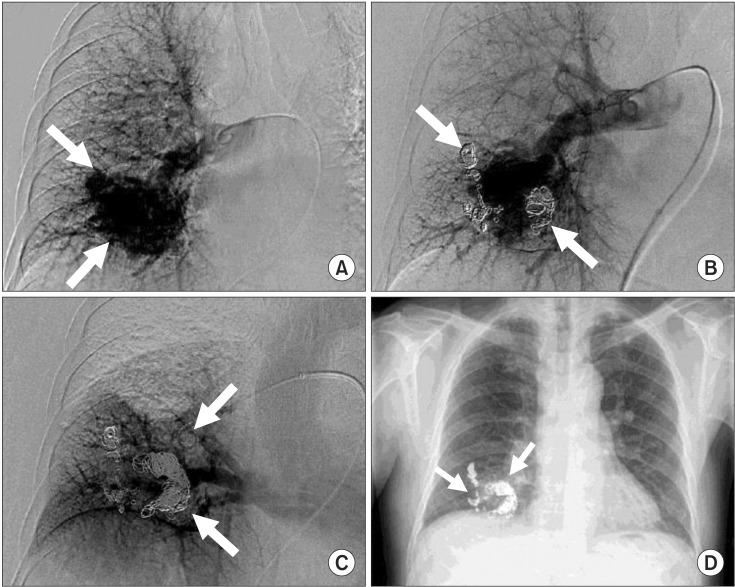
Although the size of AVM was substantially large, transcatheter embolization was chosen as a primary treatment option rather than surgical resection. We thought that the risk of thoracic surgery-related pulmonary complications would be high in this patient, considering his old age. The patient underwent pulmonary angiography via the right femoral vein, and a 64-mm pulmonary AVM was visualized in the right middle lobe. Multiple small and large feeding arteries were detected. A microcatheter was advanced, and transcatheter embolization was performed on the small feeding arteries with multiple pushable microcoils (Figure 3). A total of 24 coils ranging from 3 to 10 mm in diameter were used. Most of the small feeders were successfully occluded, but residual large feeders were still noted on post-embolization angiography. Thereafter, we scheduled a staged embolization procedure to reduce the exposure to radiocontrast media. After the first embolization, the patient's PaO2 level was found to be elevated to 62.4 mmHg, using a nasal prong at 3 L/min.
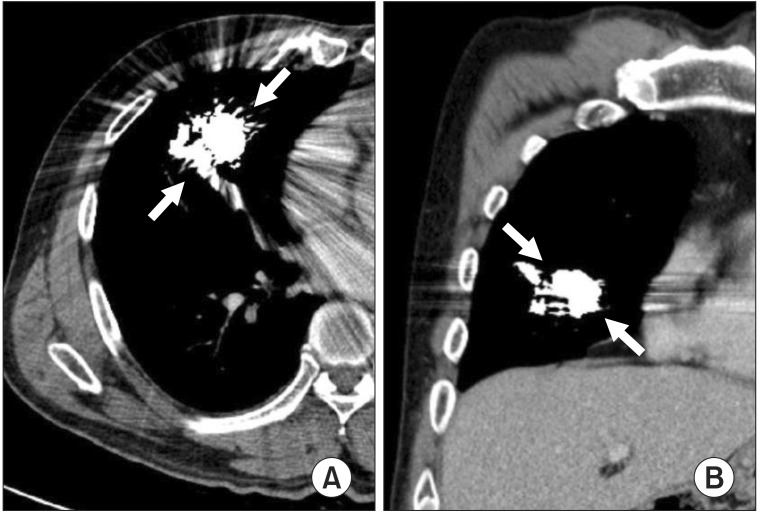
We performed a second round of embolization 2 days after the first procedure. Two large feeders were detected, and transcatheter embolization was performed with multiple interlock detachable coils (Figure 3). The diameters of the coils ranged from 8 to 14 mm, and 13 coils were deployed in total. Post-embolization angiography showed no more abnormal vascular connections, and the procedure was completed. After the second embolization procedure, his PaO2 level on room air normalized to 76.2 mm Hg. There were no procedure-related complications such as vascular access site complication or contrast induced nephropathy. There was no significant change in renal function after the procedure. The serum creatinine level was 1.06 mg/dL at admission, and 0.78 mg/dL before discharge. The patient was discharged and received a follow-up chest CT scan after 1 year, which showed no evidence of residual lesion or recanalization (Figure 4). Laboratory results also demonstrated improvement in erythrocytosis after 1 year (hemoglobin level decreased to 15.9 g/dL). The patient will undergo chest CT again after 1 year.
Pulmonary AVM is a rare disease entity. Nevertheless, it is important to consider this condition in the differential diagnosis of common pulmonary problems such as hypoxia, pulmonary nodules, or hemoptysis. Pulmonary AVM is known to be associated with HHT, which is an autosomal dominant genetic disorder. Previous epidemiologic studies reported that about 70% to 90% of pulmonary AVMs occurred in patients with HHT3. However, there is a suggestion that the association of pulmonary AVM with HHT may be lower in Asian population6. The majority of pulmonary AVMs without HHT are idiopathic, as seen in the present case.
Many patients with pulmonary AVM are reportedly asymptomatic despite having hypoxia3. This phenomenon is thought to be due to chronic compensation including the secondary erythrocytic response1. The patient in the present case also showed significant hypoxia, but denied subjective dyspnea. Despite the lack of subjective symptoms related to hypoxia, pulmonary AVMs can be associated with considerable complications. Neurological complications are the most common among these patients, including stroke, transient ischemic attack, cerebral abscess, and migraine7. Life threatening hemorrhagic complications can also occur in rare cases.
Due to the substantial morbidity rate associated with AVMs and the recent development of angiographic techniques, it is currently recommended that all pulmonary AVMs amenable to embolization should be treated with transcatheter embolization8. After successful embolization, the occlusion of feeding arteries leads to the regression of pulmonary AVMs, resulting in the resolution of right-to-left shunt, improvement to oxygenation, and the prevention of embolic complications4.
In spite of the recent advances in angiographic interventions, there are still technical limitations to embolization in some cases. It has been reported that the increasing feeding artery diameter, the use of small numbers of coils, the use of oversized coils, and proximal coil location are associated with the recanalization of pulmonary AVMs after embolization9. Large pulmonary AVMs with multiple large feeding arteries are difficult to occlude completely. Surgical resection is necessary in cases that are not amenable to transcatheter embolization. However, there is no consensus about which pulmonary AVMs should be treated with surgical resection rather than transcatheter embolization, and this choice often depends on the skill, experience, and preference of the interventionist.
There were several reports of efficacy and feasibility of transcatheter embolization for the treatment of large pulmonary AVMs10,11. An interesting and distinguishing feature of the present case is that we used a lot of coils with various sizes for a single pulmonary AVM. A total of 37 coils were utilized. This procedure was labor intensive and time consuming, but the AVM was occluded compactly without recanalization after 1 year. The present case emphasizes the importance of using multiple devices of various sizes in cases of complex AVMs to improve outcomes of transcatheter embolization.
In summary, although transcatheter embolization is the treatment of choice for most pulmonary AVMs, there are certain cases where there may be technical problems with this approach. To our knowledge, there is no clear indication or size threshold for choosing surgical resection over embolization. Considering the risks of surgery-related morbidity and mortality, transcatheter embolization should be attempted even for large AVMs. The present case demonstrates that even very large pulmonary AVMs with multiple feeding arteries can be successfully treated with repeated transcatheter embolization.
References
1. Shovlin CL. Pulmonary arteriovenous malformations. Am J Respir Crit Care Med 2014;190:1217-1228. PMID: 25420112.



2. Khurshid I, Downie GH. Pulmonary arteriovenous malformation. Postgrad Med J 2002;78:191-197. PMID: 11930021.



3. Cartin-Ceba R, Swanson KL, Krowka MJ. Pulmonary arteriovenous malformations. Chest 2013;144:1033-1044. PMID: 24008954.


4. Gossage JR, Kanj G. Pulmonary arteriovenous malformations: a state of the art review. Am J Respir Crit Care Med 1998;158:643-661. PMID: 9700146.


5. Lee DW, White RI Jr, Egglin TK, Pollak JS, Fayad PB, Wirth JA, et al. Embolotherapy of large pulmonary arteriovenous malformations: long-term results. Ann Thorac Surg 1997;64:930-939. PMID: 9354504.


6. Kim HJ, Lee JS, Oh YM, Shim TS, Lim CM, Koh YS, et al. Clinical characteristics of pulmonary arteriovenous malformations in Koreans. Respirology 2015;20:155-159. PMID: 25289945.


7. Iqbal M, Rossoff LJ, Steinberg HN, Marzouk KA, Siegel DN. Pulmonary arteriovenous malformations: a clinical review. Postgrad Med J 2000;76:390-394. PMID: 10878194.



8. Hsu CC, Kwan GN, Thompson SA, Evans-Barns H, van Driel ML. Embolisation for pulmonary arteriovenous malformation. Cochrane Database Syst Rev 2015;1:CD008017PMID: 25634560.


9. Milic A, Chan RP, Cohen JH, Faughnan ME. Reperfusion of pulmonary arteriovenous malformations after embolotherapy. J Vasc Interv Radiol 2005;16:1675-1683. PMID: 16371535.


10. Hart JL, Aldin Z, Braude P, Shovlin CL, Jackson J. Embolization of pulmonary arteriovenous malformations using the Amplatzer vascular plug: successful treatment of 69 consecutive patients. Eur Radiol 2010;20:2663-2670. PMID: 20574633.



11. Kucukay F, Ozdemir M, Senol E, Okten S, Ereren M, Karan A. Large pulmonary arteriovenous malformations: long-term results of embolization with AMPLATZER vascular plugs. J Vasc Interv Radiol 2014;25:1327-1332. PMID: 24656179.


Figure┬Ā2
(A-D) Chest computed tomography showing a huge entangled enhanced lesion in the right middle lobe, which is consistent with pulmonary arteriovenous malformation (arrows).



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Print
Print Download Citation
Download Citation




